Oncology Software Update Compliant with New BTS Guidelines
|
By MedImaging International staff writers Posted on 24 Aug 2015 |
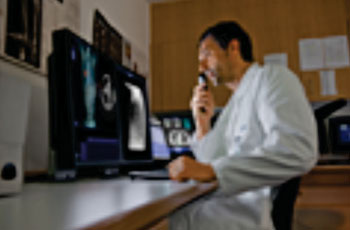
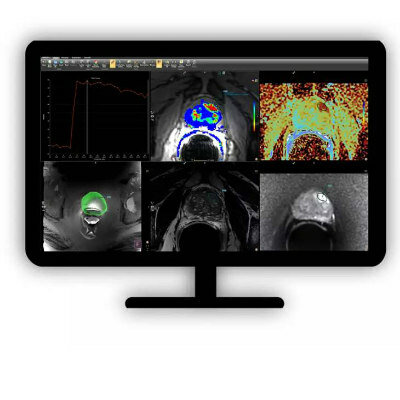
Image: Siemens Healthcare’s syngo.via MM Oncology facilitates compliance by clinicians to British Thoracic Society guidelines for the investigation and management of pulmonary nodules.(Photo courtesy of Siemens Healthcare).
A key diagnostic imaging vendor has announced that it has modified its oncology software solution to ensure compliance with new British Thoracic Society (BTS) guidelines for lung nodule screening.
The BTS developed the new evidence-based algorithms and recommendations to save clinicians time and improve outcomes for lung cancer patients. Improved estimations of the size and growth rate of lung nodules are vital in helping oncologists understand whether a nodule is cancerous, and to help them decide on treatment options. According to Cancer Research UK (London, UK) lung cancer is the second most common cancer in the UK, accounting for 13% of all new cases of cancer.
The syngo.via MM Oncology software solution was developed by Siemens Healthcare (Erlangen, Germany) and should help oncologists comply with key areas of the new guidelines. According to the guidelines, automated or semi-automated volumetric measurement is the preferred method for assessing coin lesions, and is more accurate than diameter measurements. Siemens Healthcare’s software solution can help clinicians measure nodule volumes, and quantify growth rates or reduction of nodules between the initial and the follow up scans.
Greg Baker, lead applications specialist User Services and syngo.via at Siemens Healthcare, said, “As standard, all of our customers using syngo.via are provided with a range of fully automated tools specifically designed to support clinicians in the detection, segmentation and evaluation of suspicious lesions including dedicated tools for the volumetric measurement of pulmonary nodules. The guidelines from BTS are a welcome change to ensure accuracy in measurement and evaluation. We are looking forward to working with our customers to assist with guideline compliance so clinicians are able to receive the valuable information they need to make a confident diagnosis.”
Related Links:
Siemens Healthcare
Cancer Research UK
The BTS developed the new evidence-based algorithms and recommendations to save clinicians time and improve outcomes for lung cancer patients. Improved estimations of the size and growth rate of lung nodules are vital in helping oncologists understand whether a nodule is cancerous, and to help them decide on treatment options. According to Cancer Research UK (London, UK) lung cancer is the second most common cancer in the UK, accounting for 13% of all new cases of cancer.
The syngo.via MM Oncology software solution was developed by Siemens Healthcare (Erlangen, Germany) and should help oncologists comply with key areas of the new guidelines. According to the guidelines, automated or semi-automated volumetric measurement is the preferred method for assessing coin lesions, and is more accurate than diameter measurements. Siemens Healthcare’s software solution can help clinicians measure nodule volumes, and quantify growth rates or reduction of nodules between the initial and the follow up scans.
Greg Baker, lead applications specialist User Services and syngo.via at Siemens Healthcare, said, “As standard, all of our customers using syngo.via are provided with a range of fully automated tools specifically designed to support clinicians in the detection, segmentation and evaluation of suspicious lesions including dedicated tools for the volumetric measurement of pulmonary nodules. The guidelines from BTS are a welcome change to ensure accuracy in measurement and evaluation. We are looking forward to working with our customers to assist with guideline compliance so clinicians are able to receive the valuable information they need to make a confident diagnosis.”
Related Links:
Siemens Healthcare
Cancer Research UK
Latest Imaging IT News
- Interactive AI Tool Supports Explainable Lung Nodule Assessment
- Breast Imaging Software Enhances Visualization and Tissue Characterization in Challenging Cases
- New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
- Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
- AI-Based Mammography Triage Software Helps Dramatically Improve Interpretation Process
- Artificial Intelligence (AI) Program Accurately Predicts Lung Cancer Risk from CT Images
- Image Management Platform Streamlines Treatment Plans
- AI-Based Technology for Ultrasound Image Analysis Receives FDA Approval
- AI Technology for Detecting Breast Cancer Receives CE Mark Approval
- Digital Pathology Software Improves Workflow Efficiency
Channels
Radiography
view channel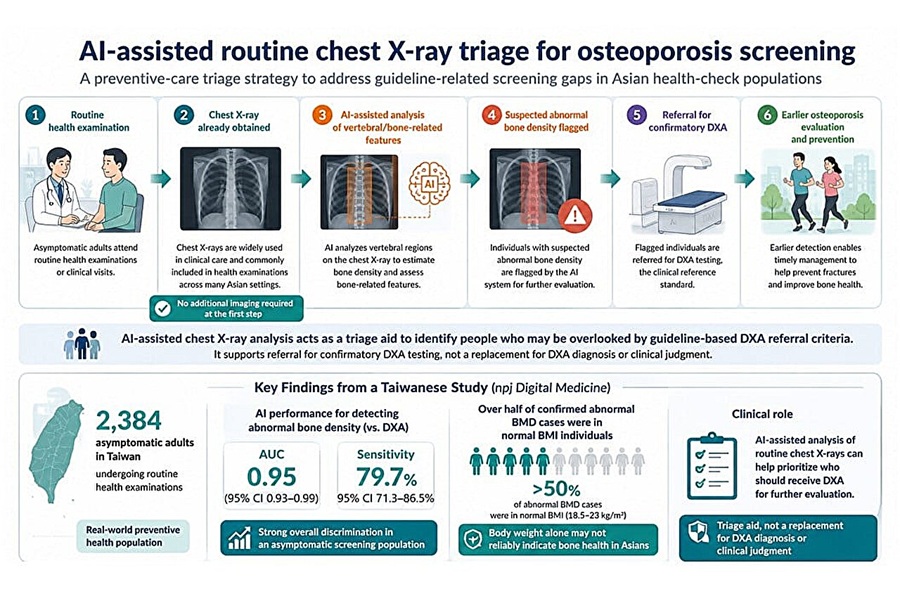
AI Tool Flags Osteoporosis Risk from Routine Chest X-Rays
Osteoporosis is a progressive loss of bone density that is often silent until a fracture occurs. Current screening frameworks concentrate on older women and select high-risk groups. Many men, younger adults,... Read more
Simple Chest X-Ray Measure Predicts Survival After Lung Cancer Surgery
Obstructive ventilatory disorder, marked by airflow limitation that reduces breathing efficiency, increases postoperative risk in patients with lung cancer. Although surgery offers the best chance of cure,... Read moreMRI
view channel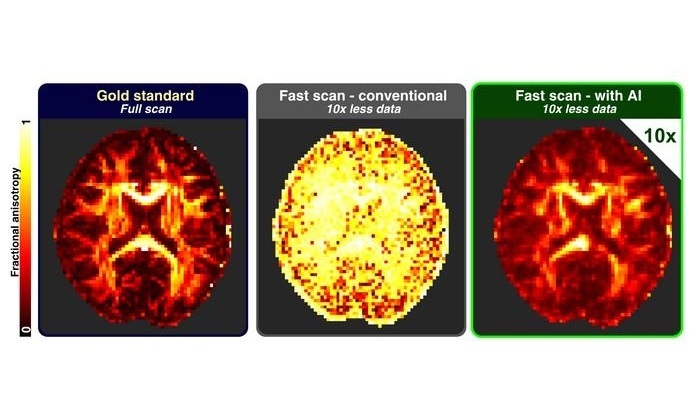
AI Approach Could Shorten Advanced Brain MRI Scans by Up to 90%
Long acquisition times for advanced brain magnetic resonance imaging (MRI) can limit access, extend waiting lists, and disrupt clinical workflows. Reducing data requirements without sacrificing image fidelity... Read more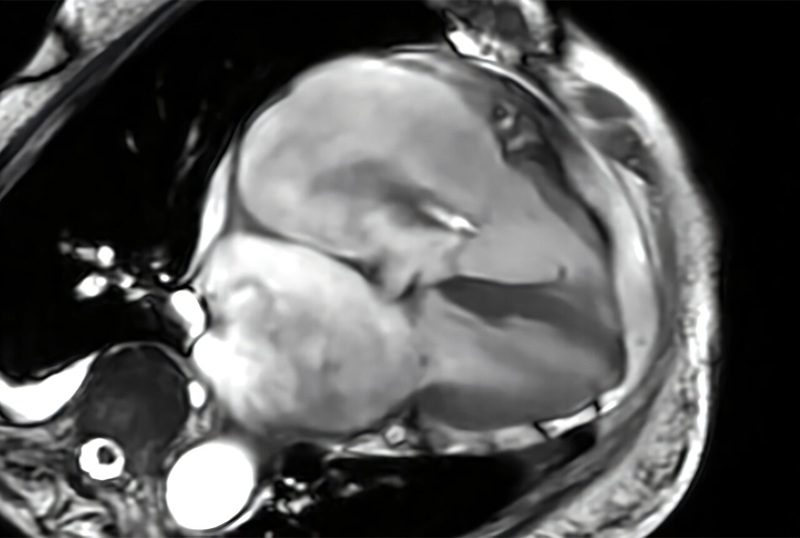
Cardiac MRI Measure Improves Risk Prediction in Tricuspid Regurgitation
Tricuspid regurgitation, in which blood flows back from the right ventricle into the right atrium, can lead to progressive right-sided heart failure. Clinicians need reliable ways to gauge severity and... Read moreUltrasound
view channelAI Robotic Ultrasound System Automates Echocardiography and Improves Consistency
Echocardiography, an ultrasound examination of the heart, is central to diagnosing and managing cardiovascular disease. Many services struggle with limited availability of skilled sonographers, variable... Read more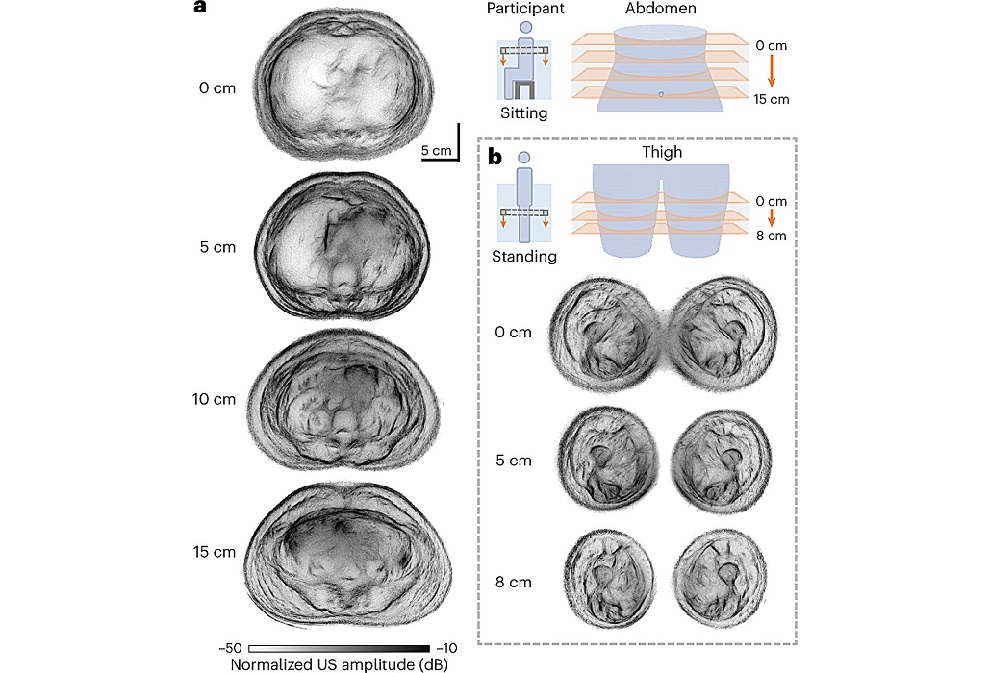
Whole Cross-Section Ultrasound System Enables Operator-Independent Imaging
Conventional ultrasound is central to bedside imaging but is limited by a narrow field of view and operator variability. Comprehensive cross-sectional assessment typically requires computed tomography... Read moreNuclear Medicine
view channel
Portable PET System Enables Real-Time Bedside Guidance for Biopsies and Ablations
Interventional radiology procedures typically rely on ultrasound, X-ray fluoroscopy, or computed tomography for image guidance. These modalities visualize anatomy but offer limited molecular information,... Read more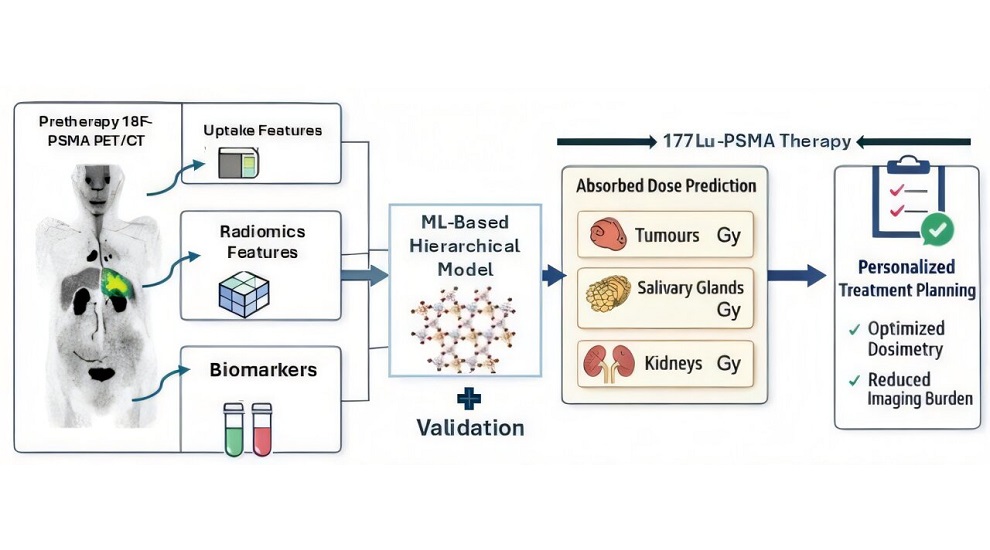
AI Model Predicts Radiation Dose Before Prostate Cancer Therapy
Metastatic castration-resistant prostate cancer (mCRPC) is an advanced form of disease that progresses despite androgen-deprivation therapy and frequently spreads to bone and visceral organs.... Read moreGeneral/Advanced Imaging
view channel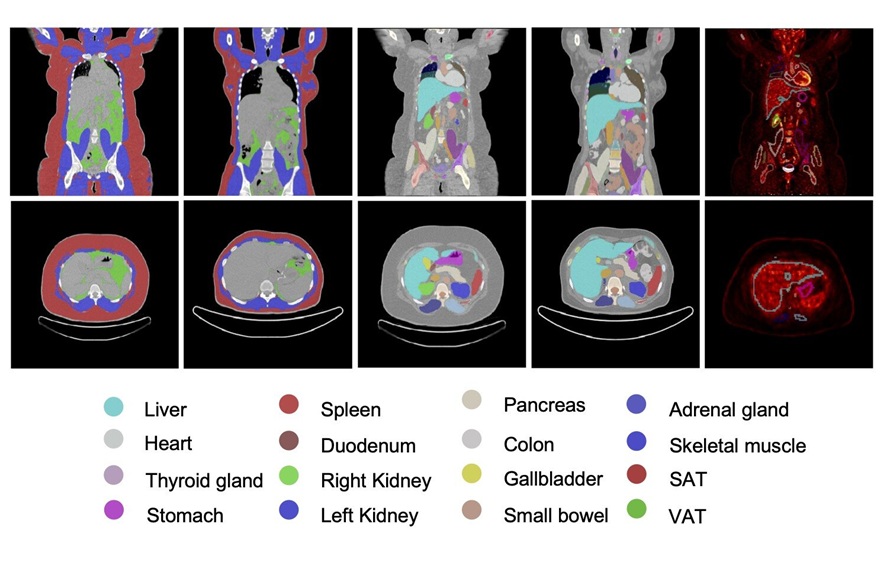
Whole-Body PET/CT Tracks Metabolic Changes After Bariatric Surgery
Obesity surgery improves weight and comorbidity profiles, yet clinicians lack tools to monitor organ-level metabolic recovery after the procedure. A clear view of systemic changes could refine follow-up... Read more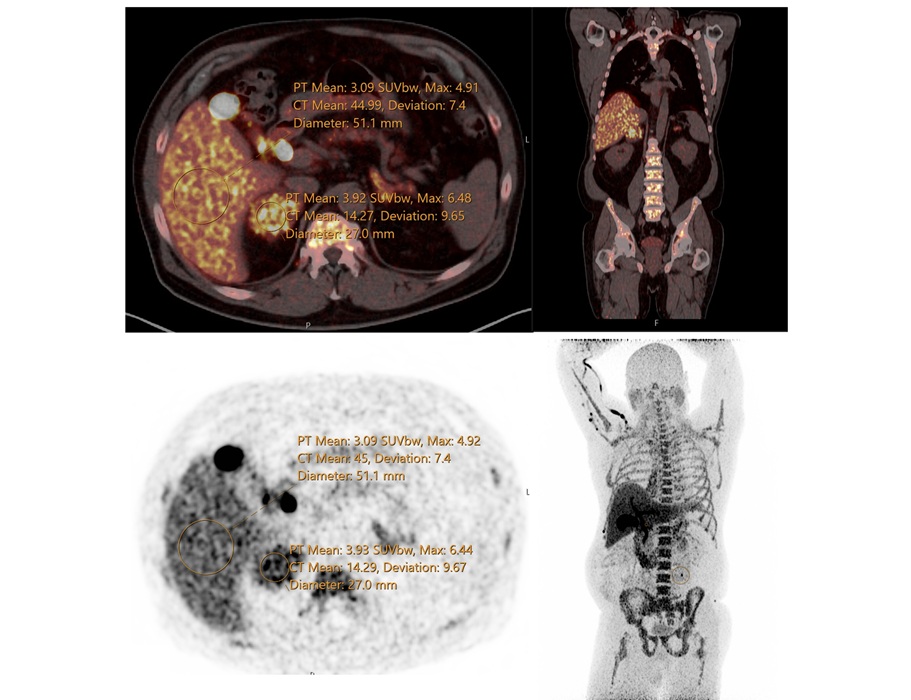
PET Tracer Localizes Overactive Adrenal Glands in Primary Aldosteronism
Primary aldosteronism (Conn’s syndrome) is the leading cause of curable secondary hypertension and results from excess aldosterone produced by the adrenal cortex. Determining whether hormone overproduction... Read moreIndustry News
view channel
GE HealthCare Showcases AI-Enabled Nuclear Medicine Portfolio at SNMMI 2026
Nuclear medicine is expanding rapidly as health systems adopt theranostics and broaden access to radiopharmaceuticals, increasing demand for scalable operations and consistent diagnostic confidence.... Read more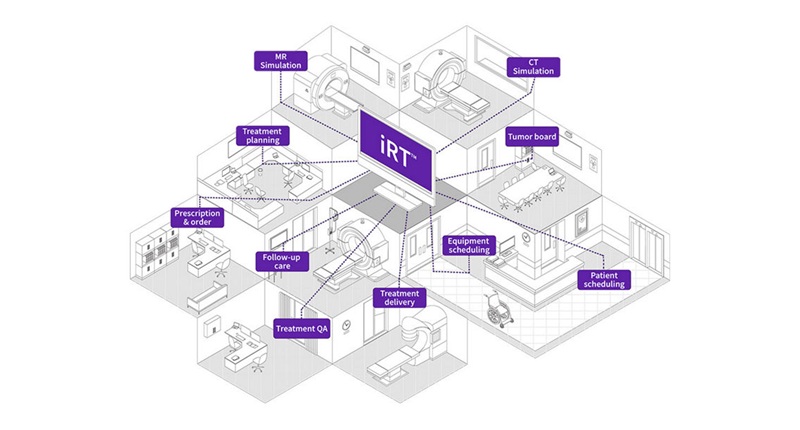
GE HealthCare Highlights AI-Supported Radiation Therapy Tools at ESTRO 2026
At the European Society for Radiotherapy and Oncology (ESTRO) 2026 Congress in Stockholm, GE HealthCare is highlighting Intelligent Radiation Therapy (iRT), MIM Software innovations, and BK Medical surgical... Read more