Combination Treatment Offers Better Option for Colon Cancer Patients
|
By MedImaging International staff writers Posted on 05 Jan 2022 |

Image: TheraSphere microscopic glass beads containing yttrium-90 (Photo courtesy of Boston Scientific)
Targeted radioembolization (TARE) alongside chemotherapy improves progress-free survival for patients with colorectal liver metastases (CLM), according to a new study.
Researchers at Northwestern University (NU; Evanston, IL, USA), the University of California, Los Angeles (UCLA; USA), and other institutions conducted an international, multicenter, open-label phase III trial of 428 patients with CLM randomly assigned to receive second-line systemic chemotherapy (with or without transarterial Yttrium-90 TARE) on a 1:1 basis. The two primary end points were progression-free survival (PFS) and hepatic PFS (hPFS), assessed by blinded independent central review.
The results showed that the patients receiving a combination of chemotherapy and TARE had longer progression-free survival, but did not improve overall survival. Some subsets of patients had greater benefit from TARE than others; characteristics which could identify patients who could benefit from addition of TARE were the location of the original colon tumor, genetic make-up of the tumor, and the amount of tumor in the liver. Importantly, the addition of TARE did not impact ability to receive subsequent therapy. The study was published on December 10, 2021, in the Journal of Clinical Oncology.
“Most patients with CLM are poor candidates for resection surgery. The cancer is either distributed in a way that it can't be operated on safely, or the size of the lesions makes it impossible,” said lead author professor Mary Mulcahy, MD, of NU. “We know systemic chemotherapy will ultimately fail, so we're looking for non-surgical therapy that can address these patients. This new treatment could be a better option compared to chemotherapy alone.”
TARE involves glass microspheres, within which a radioactive isotope (yttrium-90) is embedded. As healthy liver tissue is mainly perfused by the portal vein, TARE can be selectively administered to the tumors. The microspheres lodge in the small vessels of the tumor (embolization), emitting localized radiation therapy to targeted segments, results in tissue necrosis. The yttrium-90, with a half-life of 3.85 days, decays to Zirconium-90.
Related Links:
Northwestern University
University of California, Los Angeles
Researchers at Northwestern University (NU; Evanston, IL, USA), the University of California, Los Angeles (UCLA; USA), and other institutions conducted an international, multicenter, open-label phase III trial of 428 patients with CLM randomly assigned to receive second-line systemic chemotherapy (with or without transarterial Yttrium-90 TARE) on a 1:1 basis. The two primary end points were progression-free survival (PFS) and hepatic PFS (hPFS), assessed by blinded independent central review.
The results showed that the patients receiving a combination of chemotherapy and TARE had longer progression-free survival, but did not improve overall survival. Some subsets of patients had greater benefit from TARE than others; characteristics which could identify patients who could benefit from addition of TARE were the location of the original colon tumor, genetic make-up of the tumor, and the amount of tumor in the liver. Importantly, the addition of TARE did not impact ability to receive subsequent therapy. The study was published on December 10, 2021, in the Journal of Clinical Oncology.
“Most patients with CLM are poor candidates for resection surgery. The cancer is either distributed in a way that it can't be operated on safely, or the size of the lesions makes it impossible,” said lead author professor Mary Mulcahy, MD, of NU. “We know systemic chemotherapy will ultimately fail, so we're looking for non-surgical therapy that can address these patients. This new treatment could be a better option compared to chemotherapy alone.”
TARE involves glass microspheres, within which a radioactive isotope (yttrium-90) is embedded. As healthy liver tissue is mainly perfused by the portal vein, TARE can be selectively administered to the tumors. The microspheres lodge in the small vessels of the tumor (embolization), emitting localized radiation therapy to targeted segments, results in tissue necrosis. The yttrium-90, with a half-life of 3.85 days, decays to Zirconium-90.
Related Links:
Northwestern University
University of California, Los Angeles
Latest Nuclear Medicine News
- New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
- New Radiotheranostic System Detects and Treats Ovarian Cancer Noninvasively
- AI System Automatically and Reliably Detects Cardiac Amyloidosis Using Scintigraphy Imaging
- Early 30-Minute Dynamic FDG-PET Acquisition Could Halve Lung Scan Times
- New Method for Triggering and Imaging Seizures to Help Guide Epilepsy Surgery
- Radioguided Surgery Accurately Detects and Removes Metastatic Lymph Nodes in Prostate Cancer Patients
- New PET Tracer Detects Inflammatory Arthritis Before Symptoms Appear
- Novel PET Tracer Enhances Lesion Detection in Medullary Thyroid Cancer
- Targeted Therapy Delivers Radiation Directly To Cells in Hard-To-Treat Cancers
- New PET Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Algorithm Predicts Prostate Cancer Recurrence in Patients Treated by Radiation Therapy
- Novel PET Imaging Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Ultrafast Laser Technology to Improve Cancer Treatment
- Low-Dose Radiation Therapy Demonstrates Potential for Treatment of Heart Failure
- New PET Radiotracer Aids Early, Noninvasive Detection of Inflammatory Bowel Disease
- Combining Amino Acid PET and MRI Imaging to Help Treat Aggressive Brain Tumors
Channels
Radiography
view channel
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more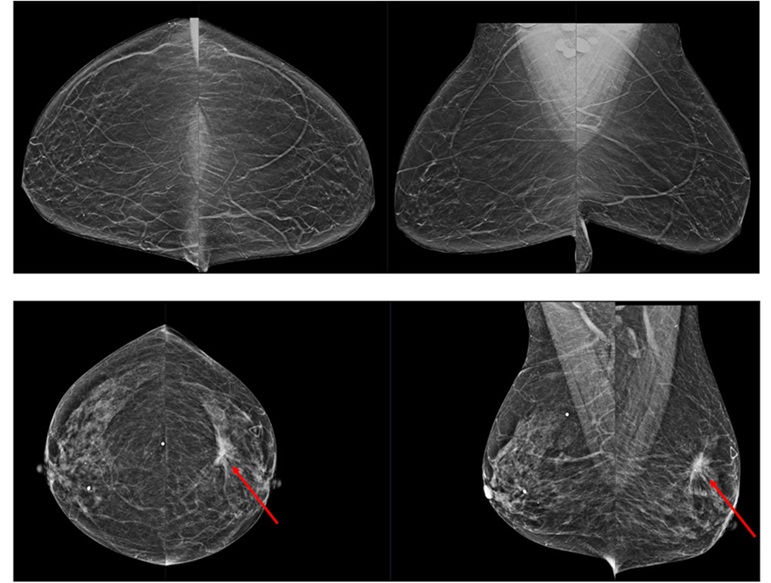
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreMRI
view channel
Diamond Dust Could Offer New Contrast Agent Option for Future MRI Scans
Gadolinium, a heavy metal used for over three decades as a contrast agent in medical imaging, enhances the clarity of MRI scans by highlighting affected areas. Despite its utility, gadolinium not only... Read more.jpg)
Combining MRI with PSA Testing Improves Clinical Outcomes for Prostate Cancer Patients
Prostate cancer is a leading health concern globally, consistently being one of the most common types of cancer among men and a major cause of cancer-related deaths. In the United States, it is the most... Read more
PET/MRI Improves Diagnostic Accuracy for Prostate Cancer Patients
The Prostate Imaging Reporting and Data System (PI-RADS) is a five-point scale to assess potential prostate cancer in MR images. PI-RADS category 3 which offers an unclear suggestion of clinically significant... Read more
Next Generation MR-Guided Focused Ultrasound Ushers In Future of Incisionless Neurosurgery
Essential tremor, often called familial, idiopathic, or benign tremor, leads to uncontrollable shaking that significantly affects a person’s life. When traditional medications do not alleviate symptoms,... Read moreUltrasound
view channel.jpg)
Groundbreaking Technology Enables Precise, Automatic Measurement of Peripheral Blood Vessels
The current standard of care of using angiographic information is often inadequate for accurately assessing vessel size in the estimated 20 million people in the U.S. who suffer from peripheral vascular disease.... Read more
Deep Learning Advances Super-Resolution Ultrasound Imaging
Ultrasound localization microscopy (ULM) is an advanced imaging technique that offers high-resolution visualization of microvascular structures. It employs microbubbles, FDA-approved contrast agents, injected... Read more
Novel Ultrasound-Launched Targeted Nanoparticle Eliminates Biofilm and Bacterial Infection
Biofilms, formed by bacteria aggregating into dense communities for protection against harsh environmental conditions, are a significant contributor to various infectious diseases. Biofilms frequently... Read moreGeneral/Advanced Imaging
view channel
PET Scans Reveal Hidden Inflammation in Multiple Sclerosis Patients
A key challenge for clinicians treating patients with multiple sclerosis (MS) is that after a certain amount of time, they continue to worsen even though their MRIs show no change. A new study has now... Read more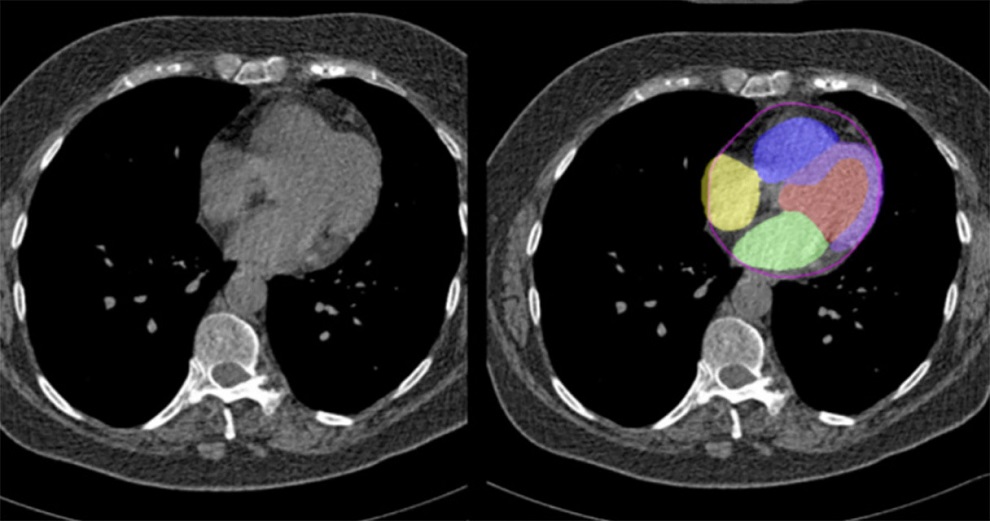
Artificial Intelligence Evaluates Cardiovascular Risk from CT Scans
Chest computed tomography (CT) is a common diagnostic tool, with approximately 15 million scans conducted each year in the United States, though many are underutilized or not fully explored.... Read more
New AI Method Captures Uncertainty in Medical Images
In the field of biomedicine, segmentation is the process of annotating pixels from an important structure in medical images, such as organs or cells. Artificial Intelligence (AI) models are utilized to... Read more.jpg)
CT Coronary Angiography Reduces Need for Invasive Tests to Diagnose Coronary Artery Disease
Coronary artery disease (CAD), one of the leading causes of death worldwide, involves the narrowing of coronary arteries due to atherosclerosis, resulting in insufficient blood flow to the heart muscle.... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more