Autofluorescence Imaging Helps Identify Radiation-Resistant Tumors
|
By MedImaging International staff writers Posted on 09 Jul 2018 |
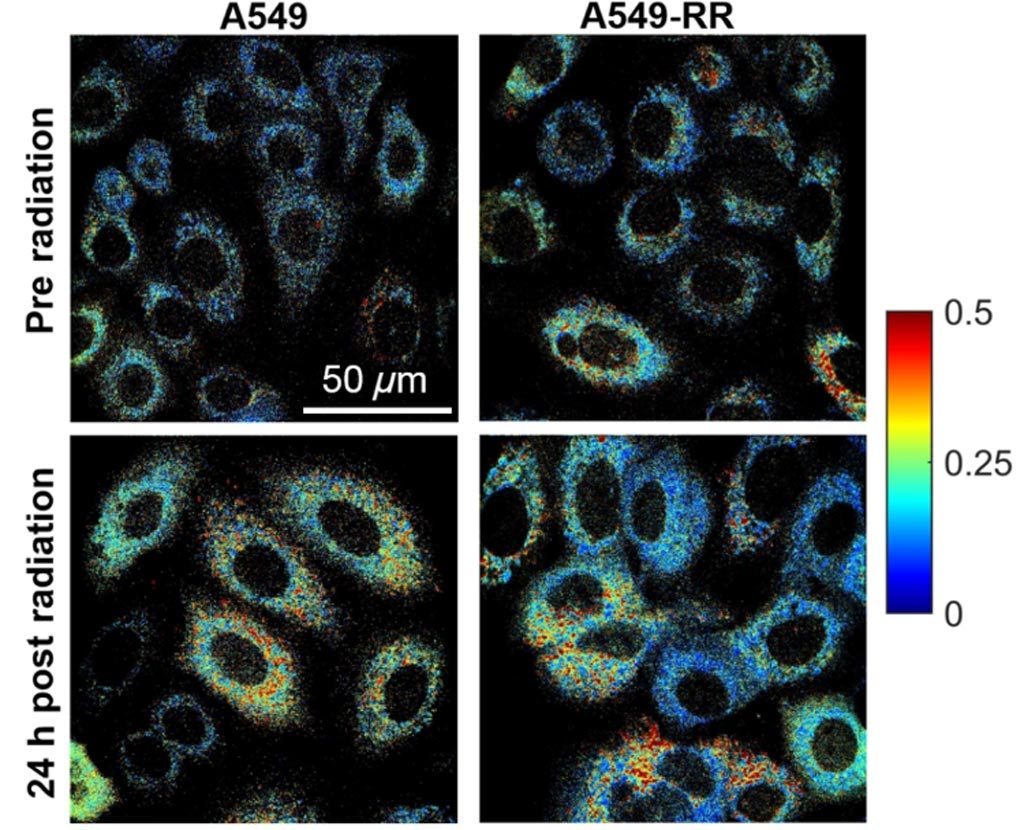
Image: Cell metabolism changes reflected in autofluorescence imaging (Photo courtesy of University of Arkansas).
An innovative imaging system can differentiate between the metabolic response of radiation-resistant and radiation-sensitive lung cancer cells, according to a new study.
Researchers at the University of Arkansas (Fayetteville, USA) and the University of Arkansas for Medical Sciences (UAMS; Little Rock, USA) used autofluorescence imaging to observe changes in nicotinamide adenine dinucleotide (NADH) and flavin adenine dinucleotide (FAD), two fluorescent molecules that play a critical role in metabolic pathways within cells responsible for respiration and energy production. To do so, they studied a radiation-resistant isogenic line of human A549 lung cancer cells which expresses a significantly elevated level of hypoxia-inducible factor (HIF-1α) and increased glucose catabolism compared with the parental, radiation-sensitive cell line.
By measuring the relative contributions of NADH and FAD to these processes, the researchers hoped to evaluate biochemical details related to cell metabolism. They therefore exposed the A549 cells to both radiation and the chemotherapy drug YC-1, a potent HIF-1α inhibitor. With the aid of fluorescence microscopy, they determined changes in the optical redox ratio of FAD/(NADH+FAD) over a period of 24 hours following treatment with YC-1, radiation, and both radiation and YC-1. They also evaluated changes in mitochondrial organization, glucose uptake, reactive oxygen species (ROS), and reduced glutathione.
They found significant differences in the optical redox ratio of radiation-resistant and sensitive A549 cells in response to radiation or YC-1 treatment alone; however, the combined treatment eliminated these differences. According to the researchers, the results show that optical redox ratio can reveal radiosensitization of previously radiation-resistant A549 cancer cells, and also provide a method for evaluating treatment response in patient-derived tumor biopsies. The study was published on June 11, 2018, in Scientific Reports.
“The use of autofluorescence imaging of cell metabolism can identify treatment-resistant cancer cells. More importantly, we think that this technique provides a sound method to evaluate tumor response to treatment and match tumors to the right therapy,” said senior author biomedical engineer Narasimhan Rajaram, PhD, of the University of Arkansas. “Non-destructive, label-free imaging approach is a valuable technique for characterizing metabolic reprogramming, and has potential clinical application to identify treatment efficacy in tumor-derived organoids.”
Hypoxic tumors tend to respond poorly to radiation because DNA damage relies on the presence of oxygen. As a regulator of oxygen homeostasis, HIF-1α plays a key role in downregulating mitochondrial oxygen consumption and enhancing transcription of important glycolytic genes; targeting HIF-1α has therefor shown promise in sensitizing radiation-resistant cancer cells to radiotherapy. Other factors in the tumor microenvironment, such as poor oxygen perfusion, can also contribute to elevated levels of HIF-1α, thereby compounding the cellular response to radiation.
Related Links:
University of Arkansas
University of Arkansas for Medical Sciences
Researchers at the University of Arkansas (Fayetteville, USA) and the University of Arkansas for Medical Sciences (UAMS; Little Rock, USA) used autofluorescence imaging to observe changes in nicotinamide adenine dinucleotide (NADH) and flavin adenine dinucleotide (FAD), two fluorescent molecules that play a critical role in metabolic pathways within cells responsible for respiration and energy production. To do so, they studied a radiation-resistant isogenic line of human A549 lung cancer cells which expresses a significantly elevated level of hypoxia-inducible factor (HIF-1α) and increased glucose catabolism compared with the parental, radiation-sensitive cell line.
By measuring the relative contributions of NADH and FAD to these processes, the researchers hoped to evaluate biochemical details related to cell metabolism. They therefore exposed the A549 cells to both radiation and the chemotherapy drug YC-1, a potent HIF-1α inhibitor. With the aid of fluorescence microscopy, they determined changes in the optical redox ratio of FAD/(NADH+FAD) over a period of 24 hours following treatment with YC-1, radiation, and both radiation and YC-1. They also evaluated changes in mitochondrial organization, glucose uptake, reactive oxygen species (ROS), and reduced glutathione.
They found significant differences in the optical redox ratio of radiation-resistant and sensitive A549 cells in response to radiation or YC-1 treatment alone; however, the combined treatment eliminated these differences. According to the researchers, the results show that optical redox ratio can reveal radiosensitization of previously radiation-resistant A549 cancer cells, and also provide a method for evaluating treatment response in patient-derived tumor biopsies. The study was published on June 11, 2018, in Scientific Reports.
“The use of autofluorescence imaging of cell metabolism can identify treatment-resistant cancer cells. More importantly, we think that this technique provides a sound method to evaluate tumor response to treatment and match tumors to the right therapy,” said senior author biomedical engineer Narasimhan Rajaram, PhD, of the University of Arkansas. “Non-destructive, label-free imaging approach is a valuable technique for characterizing metabolic reprogramming, and has potential clinical application to identify treatment efficacy in tumor-derived organoids.”
Hypoxic tumors tend to respond poorly to radiation because DNA damage relies on the presence of oxygen. As a regulator of oxygen homeostasis, HIF-1α plays a key role in downregulating mitochondrial oxygen consumption and enhancing transcription of important glycolytic genes; targeting HIF-1α has therefor shown promise in sensitizing radiation-resistant cancer cells to radiotherapy. Other factors in the tumor microenvironment, such as poor oxygen perfusion, can also contribute to elevated levels of HIF-1α, thereby compounding the cellular response to radiation.
Related Links:
University of Arkansas
University of Arkansas for Medical Sciences
Latest Nuclear Medicine News
- New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
- New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
- New Radiotheranostic System Detects and Treats Ovarian Cancer Noninvasively
- AI System Automatically and Reliably Detects Cardiac Amyloidosis Using Scintigraphy Imaging
- Early 30-Minute Dynamic FDG-PET Acquisition Could Halve Lung Scan Times
- New Method for Triggering and Imaging Seizures to Help Guide Epilepsy Surgery
- Radioguided Surgery Accurately Detects and Removes Metastatic Lymph Nodes in Prostate Cancer Patients
- New PET Tracer Detects Inflammatory Arthritis Before Symptoms Appear
- Novel PET Tracer Enhances Lesion Detection in Medullary Thyroid Cancer
- Targeted Therapy Delivers Radiation Directly To Cells in Hard-To-Treat Cancers
- New PET Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Algorithm Predicts Prostate Cancer Recurrence in Patients Treated by Radiation Therapy
- Novel PET Imaging Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Ultrafast Laser Technology to Improve Cancer Treatment
- Low-Dose Radiation Therapy Demonstrates Potential for Treatment of Heart Failure
- New PET Radiotracer Aids Early, Noninvasive Detection of Inflammatory Bowel Disease
Channels
Radiography
view channel
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreMRI
view channel
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read more
World's First Sensor Detects Errors in MRI Scans Using Laser Light and Gas
MRI scanners are daily tools for doctors and healthcare professionals, providing unparalleled 3D imaging of the brain, vital organs, and soft tissues, far surpassing other imaging technologies in quality.... Read more
Diamond Dust Could Offer New Contrast Agent Option for Future MRI Scans
Gadolinium, a heavy metal used for over three decades as a contrast agent in medical imaging, enhances the clarity of MRI scans by highlighting affected areas. Despite its utility, gadolinium not only... Read more.jpg)
Combining MRI with PSA Testing Improves Clinical Outcomes for Prostate Cancer Patients
Prostate cancer is a leading health concern globally, consistently being one of the most common types of cancer among men and a major cause of cancer-related deaths. In the United States, it is the most... Read moreUltrasound
view channel
First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity
Ultrasound scans are essential for identifying and diagnosing various medical conditions, but often, patients must wait weeks or months for results due to a shortage of qualified medical professionals... Read more
Largest Model Trained On Echocardiography Images Assesses Heart Structure and Function
Foundation models represent an exciting frontier in generative artificial intelligence (AI), yet many lack the specialized medical data needed to make them applicable in healthcare settings.... Read more.jpg)
Groundbreaking Technology Enables Precise, Automatic Measurement of Peripheral Blood Vessels
The current standard of care of using angiographic information is often inadequate for accurately assessing vessel size in the estimated 20 million people in the U.S. who suffer from peripheral vascular disease.... Read moreGeneral/Advanced Imaging
view channel
Radiation Therapy Computed Tomography Solution Boosts Imaging Accuracy
One of the most significant challenges in oncology care is disease complexity in terms of the variety of cancer types and the individualized presentation of each patient. This complexity necessitates a... Read more
PET Scans Reveal Hidden Inflammation in Multiple Sclerosis Patients
A key challenge for clinicians treating patients with multiple sclerosis (MS) is that after a certain amount of time, they continue to worsen even though their MRIs show no change. A new study has now... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more