US FDA Approves First Injectable Intravenous Suspension for Contrast-Enhanced Liver Ultrasonography
|
By MedImaging International staff writers Posted on 11 Apr 2016 |
A leading global diagnostic imaging company has announced US FDA approval of injectable contrast agent for the characterization of focal liver lesions, using ultrasonography of the liver.
The US Food and Drug Administration (FDA; Silver Spring, MD USA) approved the contrast agent, “sulfur hexafluoride lipid-type A microspheres,” for use in adults with suboptimal echocardiograms, and also for children. This is the first ultrasound contrast agent approved by US FDA approval for use in liver imaging, and the first one approved for pediatric patients. The agent improves the sensitivity, and specificity of ultrasonography, and helps surgeons differentiate between focal hepatic lesions that are benign and those that are malignant.
The LUMASON agent, also called SonoVue, was developed by Bracco Imaging (Monroe Township, NJ, USA), and is already being used in Europe and Asia. The agent consists of gas-filled microspheres which reflect sound waves, and enhance the ultrasonographic images. The system has a proven safety, and efficacy profile for liver ultrasonography, and echocardiography, and is packaged in a three-part kit that does not need mechanical agitation, or refrigeration.
Alberto Spinazzi, MD, senior VP, global medical and regulatory affairs, in the Bracco group, said, “We are proud to be the first company to obtain FDA approval for this important clinical use of ultrasound contrast both in adults and children. This new indication for LUMASON reflects our efforts and investments to expand the range of approved clinical indications for contrast enhanced ultrasound in the United States. We are very pleased with the collaborative work with the FDA whose review is critical to ensuring the safety and efficacy of any use of imaging products to the healthcare community.”
Related Links:
FDA
Bracco
The US Food and Drug Administration (FDA; Silver Spring, MD USA) approved the contrast agent, “sulfur hexafluoride lipid-type A microspheres,” for use in adults with suboptimal echocardiograms, and also for children. This is the first ultrasound contrast agent approved by US FDA approval for use in liver imaging, and the first one approved for pediatric patients. The agent improves the sensitivity, and specificity of ultrasonography, and helps surgeons differentiate between focal hepatic lesions that are benign and those that are malignant.
The LUMASON agent, also called SonoVue, was developed by Bracco Imaging (Monroe Township, NJ, USA), and is already being used in Europe and Asia. The agent consists of gas-filled microspheres which reflect sound waves, and enhance the ultrasonographic images. The system has a proven safety, and efficacy profile for liver ultrasonography, and echocardiography, and is packaged in a three-part kit that does not need mechanical agitation, or refrigeration.
Alberto Spinazzi, MD, senior VP, global medical and regulatory affairs, in the Bracco group, said, “We are proud to be the first company to obtain FDA approval for this important clinical use of ultrasound contrast both in adults and children. This new indication for LUMASON reflects our efforts and investments to expand the range of approved clinical indications for contrast enhanced ultrasound in the United States. We are very pleased with the collaborative work with the FDA whose review is critical to ensuring the safety and efficacy of any use of imaging products to the healthcare community.”
Related Links:
FDA
Bracco
Latest Ultrasound News
- Diagnostic System Automatically Analyzes TTE Images to Identify Congenital Heart Disease
- Super-Resolution Imaging Technique Could Improve Evaluation of Cardiac Conditions

- First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity

- Largest Model Trained On Echocardiography Images Assesses Heart Structure and Function
- Groundbreaking Technology Enables Precise, Automatic Measurement of Peripheral Blood Vessels
- Deep Learning Advances Super-Resolution Ultrasound Imaging
- Novel Ultrasound-Launched Targeted Nanoparticle Eliminates Biofilm and Bacterial Infection
- AI-Guided Ultrasound System Enables Rapid Assessments of DVT
- Focused Ultrasound Technique Gets Quality Assurance Protocol
- AI-Guided Handheld Ultrasound System Helps Capture Diagnostic-Quality Cardiac Images
- Non-Invasive Ultrasound Imaging Device Diagnoses Risk of Chronic Kidney Disease
- Wearable Ultrasound Platform Paves Way for 24/7 Blood Pressure Monitoring On the Wrist
- Diagnostic Ultrasound Enhancing Agent to Improve Image Quality in Pediatric Heart Patients
- AI Detects COVID-19 in Lung Ultrasound Images
- New Ultrasound Technology to Revolutionize Respiratory Disease Diagnoses
- Dynamic Contrast-Enhanced Ultrasound Highly Useful For Interventions
Channels
Radiography
view channel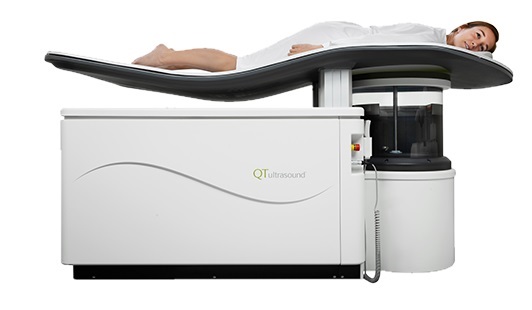
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more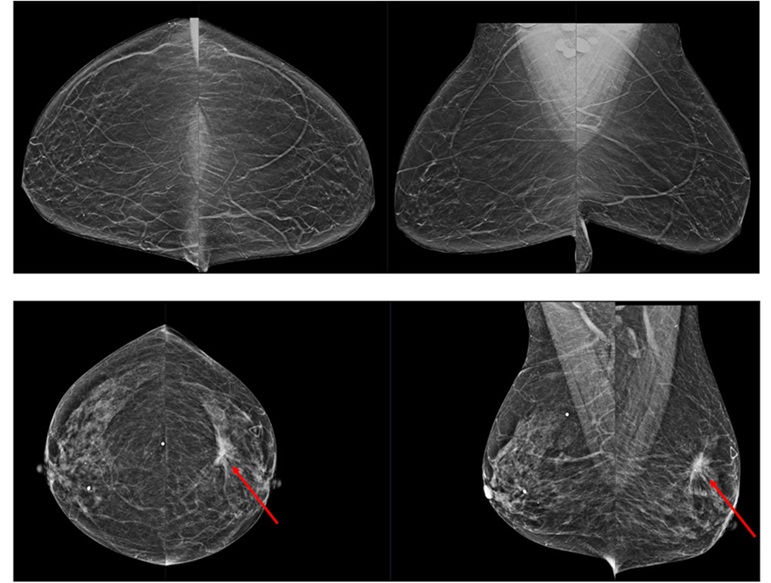
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreMRI
view channel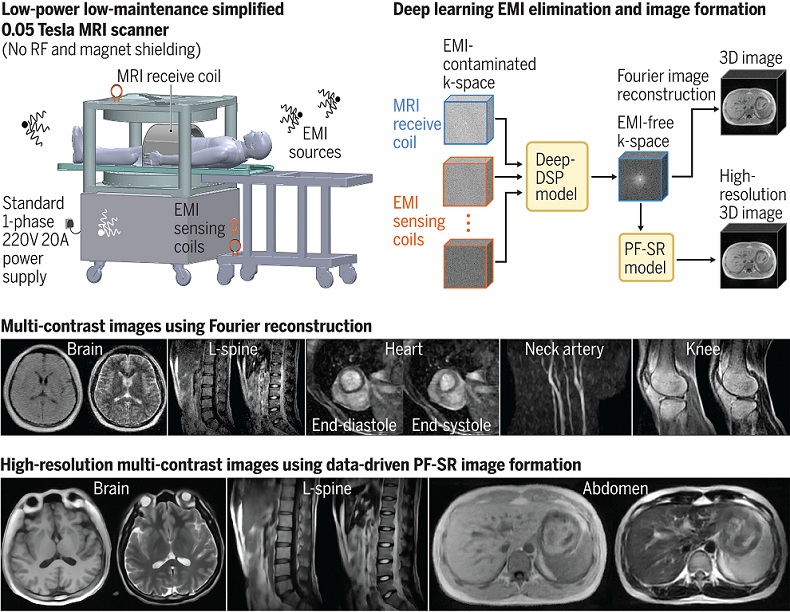
Low-Cost Whole-Body MRI Device Combined with AI Generates High-Quality Results
Magnetic Resonance Imaging (MRI) has significantly transformed healthcare, providing a noninvasive, radiation-free method for detailed imaging. It is especially promising for the future of medical diagnosis... Read more
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read moreNuclear Medicine
view channelNew PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
Clear cell renal cell cancer (ccRCC) represents 70-80% of renal cell carcinoma cases. While localized disease can be effectively treated with surgery and ablative therapies, one-third of patients either... Read more
New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
Imaging inflammation using traditional radiological techniques presents significant challenges, including radiation exposure, poor image quality, high costs, and invasive procedures. Now, new contrast... Read more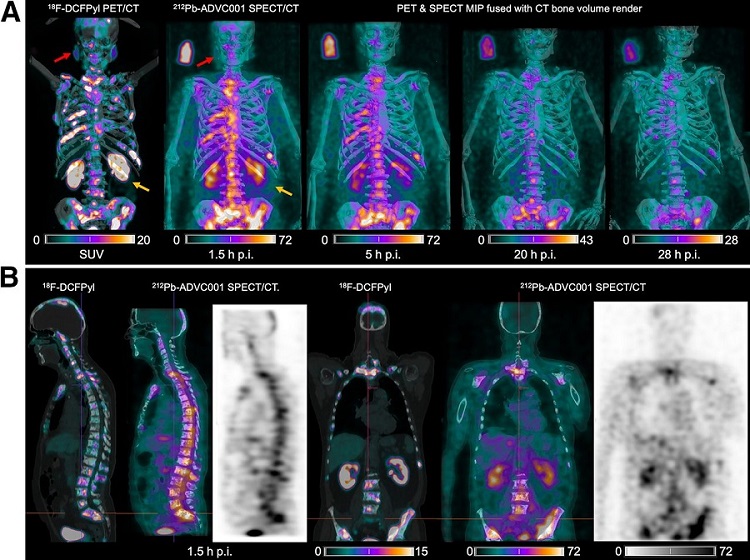
New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
The development of lead-212 (212Pb)-PSMA–based targeted alpha therapy (TAT) is garnering significant interest in treating patients with metastatic castration-resistant prostate cancer. The imaging of 212Pb,... Read moreGeneral/Advanced Imaging
view channelBone Density Test Uses Existing CT Images to Predict Fractures
Osteoporotic fractures are not only devastating and deadly, especially hip fractures, but also impose significant costs. They rank among the top chronic diseases in terms of disability-adjusted life years... Read more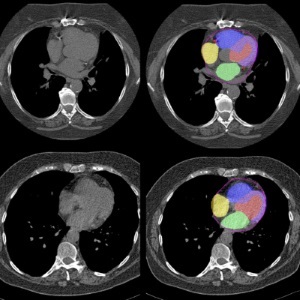
AI Predicts Cardiac Risk and Mortality from Routine Chest CT Scans
Heart disease remains the leading cause of death and is largely preventable, yet many individuals are unaware of their risk until it becomes severe. Early detection through screening can reveal heart issues,... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more