Improved Molecular Breast Imaging Screening System Receives CE Marking
|
By MedImaging International staff writers Posted on 26 Oct 2015 |
A breast imaging medical device manufacturer has announced that their Molecular Breast Imaging (MBI) system, intended for women with dense breast tissue, has received the European CE Marking.
The MBI screening system is already in use in leading hospitals in the US following approval by the US Food and Drug Administration (FDA) in 2011. The system is intended for the screening and early detection of breast cancer in women with dense breast tissue for whom conventional mammography and screening with other modalities is insufficient.
CE Marking approval of the LumaGEM MBA screening system was announced by the manufacturer, Gamma Medica (Salem, NH, USA), a women’s health company, and enables the company to sell and distribute the system in the European Economic Area (EEA).
Nearly one in 10 women in Europe develops breast cancer before the age of 80, making it the common cancer affecting women. Conventional mammography has less sensitivity when imaging dense breast tissue – both dense tissue and cancerous areas appear white on the mammogram. At the same time women with dense breast have an increased risk of developing breast cancer. A recent has shown that the addition of MBI screening test to an annual mammogram resulted in a nearly 400% percent increase in detection rates of invasive breast cancer. The use of MBI screening also resulted in 50% less biopsies, and cost savings of 15% per cancer detected, compared to only screening with conventional mammography.
MBI is a functional imaging modality and can show activity and detect tumors in dense tissue, whereas other modalities such as ultrasound, Magnetic Resonance Imaging (MRI), and tomosynthesis, are anatomical imaging systems. MBI is also useful for high-risk patients, and those who cannot undergo an MRI exam.
Philip M. Croxford, President and CEO of Gamma Medica, said, “Securing the CE Mark for the LumaGEM MBI system represents a major step forward in our commercialization plans to market this technology to benefit women with dense breast tissue in Europe. By offering the best MBI solution to physicians, we are helping them manage their patients’ risk efficiently by providing a highly accurate and cost-efficient secondary breast cancer screening for women with complex mammograms and dense breast tissue.”
Related Links:
Gamma Medica
The MBI screening system is already in use in leading hospitals in the US following approval by the US Food and Drug Administration (FDA) in 2011. The system is intended for the screening and early detection of breast cancer in women with dense breast tissue for whom conventional mammography and screening with other modalities is insufficient.
CE Marking approval of the LumaGEM MBA screening system was announced by the manufacturer, Gamma Medica (Salem, NH, USA), a women’s health company, and enables the company to sell and distribute the system in the European Economic Area (EEA).
Nearly one in 10 women in Europe develops breast cancer before the age of 80, making it the common cancer affecting women. Conventional mammography has less sensitivity when imaging dense breast tissue – both dense tissue and cancerous areas appear white on the mammogram. At the same time women with dense breast have an increased risk of developing breast cancer. A recent has shown that the addition of MBI screening test to an annual mammogram resulted in a nearly 400% percent increase in detection rates of invasive breast cancer. The use of MBI screening also resulted in 50% less biopsies, and cost savings of 15% per cancer detected, compared to only screening with conventional mammography.
MBI is a functional imaging modality and can show activity and detect tumors in dense tissue, whereas other modalities such as ultrasound, Magnetic Resonance Imaging (MRI), and tomosynthesis, are anatomical imaging systems. MBI is also useful for high-risk patients, and those who cannot undergo an MRI exam.
Philip M. Croxford, President and CEO of Gamma Medica, said, “Securing the CE Mark for the LumaGEM MBI system represents a major step forward in our commercialization plans to market this technology to benefit women with dense breast tissue in Europe. By offering the best MBI solution to physicians, we are helping them manage their patients’ risk efficiently by providing a highly accurate and cost-efficient secondary breast cancer screening for women with complex mammograms and dense breast tissue.”
Related Links:
Gamma Medica
Latest Nuclear Medicine News
- New PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
- New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
- New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
- New Radiotheranostic System Detects and Treats Ovarian Cancer Noninvasively
- AI System Automatically and Reliably Detects Cardiac Amyloidosis Using Scintigraphy Imaging
- Early 30-Minute Dynamic FDG-PET Acquisition Could Halve Lung Scan Times
- New Method for Triggering and Imaging Seizures to Help Guide Epilepsy Surgery
- Radioguided Surgery Accurately Detects and Removes Metastatic Lymph Nodes in Prostate Cancer Patients
- New PET Tracer Detects Inflammatory Arthritis Before Symptoms Appear
- Novel PET Tracer Enhances Lesion Detection in Medullary Thyroid Cancer
- Targeted Therapy Delivers Radiation Directly To Cells in Hard-To-Treat Cancers
- New PET Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Algorithm Predicts Prostate Cancer Recurrence in Patients Treated by Radiation Therapy
- Novel PET Imaging Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Ultrafast Laser Technology to Improve Cancer Treatment
- Low-Dose Radiation Therapy Demonstrates Potential for Treatment of Heart Failure
Channels
MRI
view channel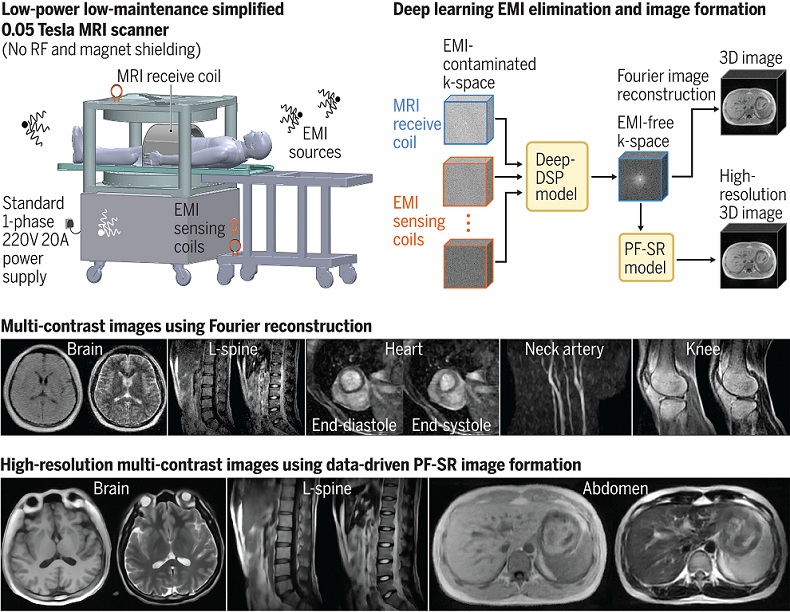
Low-Cost Whole-Body MRI Device Combined with AI Generates High-Quality Results
Magnetic Resonance Imaging (MRI) has significantly transformed healthcare, providing a noninvasive, radiation-free method for detailed imaging. It is especially promising for the future of medical diagnosis... Read more
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read moreUltrasound
view channel.jpg)
Diagnostic System Automatically Analyzes TTE Images to Identify Congenital Heart Disease
Congenital heart disease (CHD) is one of the most prevalent congenital anomalies worldwide, presenting substantial health and financial challenges for affected patients. Early detection and treatment of... Read more
Super-Resolution Imaging Technique Could Improve Evaluation of Cardiac Conditions
The heart depends on efficient blood circulation to pump blood throughout the body, delivering oxygen to tissues and removing carbon dioxide and waste. Yet, when heart vessels are damaged, it can disrupt... Read more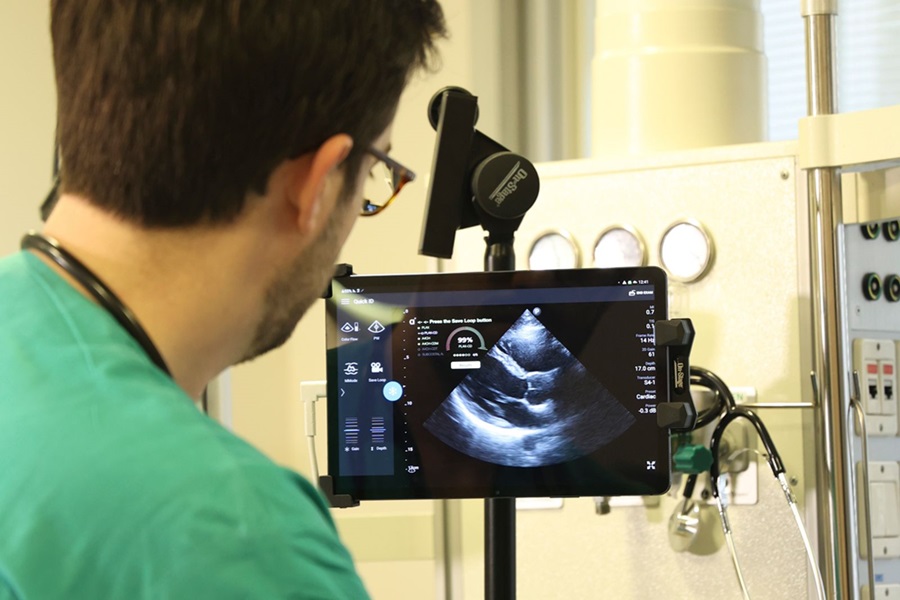
First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity
Ultrasound scans are essential for identifying and diagnosing various medical conditions, but often, patients must wait weeks or months for results due to a shortage of qualified medical professionals... Read moreNuclear Medicine
view channelNew PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
Clear cell renal cell cancer (ccRCC) represents 70-80% of renal cell carcinoma cases. While localized disease can be effectively treated with surgery and ablative therapies, one-third of patients either... Read more
New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
Imaging inflammation using traditional radiological techniques presents significant challenges, including radiation exposure, poor image quality, high costs, and invasive procedures. Now, new contrast... Read more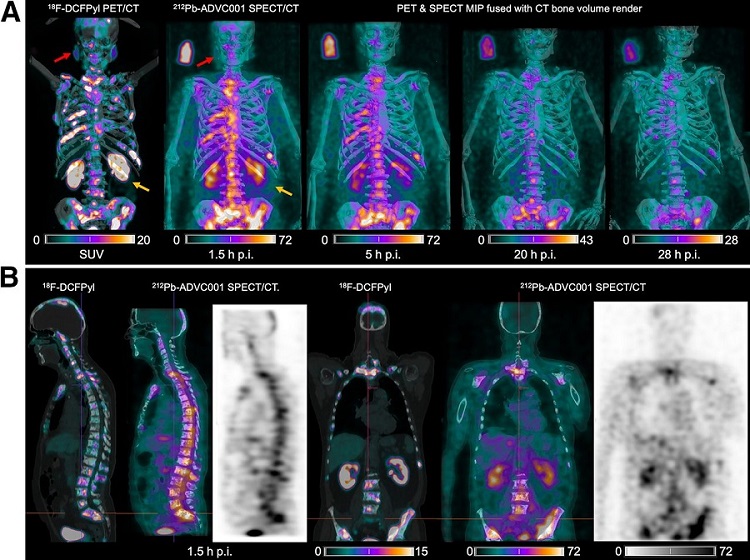
New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
The development of lead-212 (212Pb)-PSMA–based targeted alpha therapy (TAT) is garnering significant interest in treating patients with metastatic castration-resistant prostate cancer. The imaging of 212Pb,... Read moreGeneral/Advanced Imaging
view channelBone Density Test Uses Existing CT Images to Predict Fractures
Osteoporotic fractures are not only devastating and deadly, especially hip fractures, but also impose significant costs. They rank among the top chronic diseases in terms of disability-adjusted life years... Read more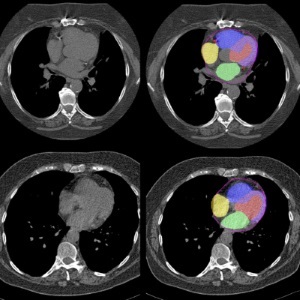
AI Predicts Cardiac Risk and Mortality from Routine Chest CT Scans
Heart disease remains the leading cause of death and is largely preventable, yet many individuals are unaware of their risk until it becomes severe. Early detection through screening can reveal heart issues,... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more