Philips Debuts First App-Based Ultrasound
|
By MedImaging International staff writers Posted on 28 Jun 2015 |
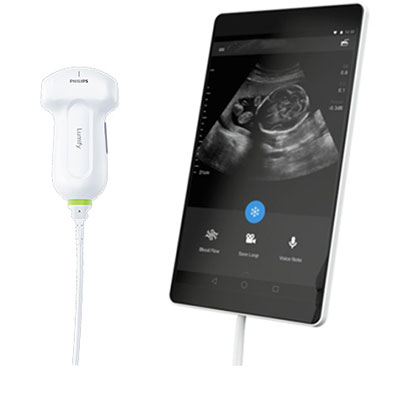
An app-based ultrasound solution aims extending the reach of ultrasound applications using mobile technology.
The app was presented at the Social Media and Critical Care conference, June, 23–26, 2015 (SMACC; Chicago, IL, USA). The app will use secure cloud-enabled technology, and offer high-image quality to a broad network of healthcare providers.
The Lumify app, was developed by Royal Philips (Amsterdam, the Netherlands) and has received US Food and Drug Administration (FDA; Silver Spring, MD USA) 510(k) clearance. The app will be available in the US later in 2015, and its introduction will be followed by the introduction of a new app-based portal, additional ultrasound transducers, applications and services.
The Lumify app will be offered through a subscription model, and provide high-quality imaging on smart devices. The app is designed for urgent care centers, emergency departments, and other clinical settings. App data will be accessible on the Philips HealthSuite Digital Platform, a cloud-based IT system.
Vitor Rocha, CEO of Philips Ultrasound, said, “Ultrasound’s versatility, portability and safety enabled it to become one of the most widely used first-line diagnostic tools. With the advent of App-Based Ultrasound, we strive to bring the value and benefits of ultrasound to more places across the health continuum. App-Based Ultrasound provides valuable information to the right people at the right time. It’s designed to drive transformation in care delivery and digital health – a dynamic combination that can extend the reach of ultrasound in a remarkable way.”
Related Links:
SMACC
Royal Philips
The app was presented at the Social Media and Critical Care conference, June, 23–26, 2015 (SMACC; Chicago, IL, USA). The app will use secure cloud-enabled technology, and offer high-image quality to a broad network of healthcare providers.
The Lumify app, was developed by Royal Philips (Amsterdam, the Netherlands) and has received US Food and Drug Administration (FDA; Silver Spring, MD USA) 510(k) clearance. The app will be available in the US later in 2015, and its introduction will be followed by the introduction of a new app-based portal, additional ultrasound transducers, applications and services.
The Lumify app will be offered through a subscription model, and provide high-quality imaging on smart devices. The app is designed for urgent care centers, emergency departments, and other clinical settings. App data will be accessible on the Philips HealthSuite Digital Platform, a cloud-based IT system.
Vitor Rocha, CEO of Philips Ultrasound, said, “Ultrasound’s versatility, portability and safety enabled it to become one of the most widely used first-line diagnostic tools. With the advent of App-Based Ultrasound, we strive to bring the value and benefits of ultrasound to more places across the health continuum. App-Based Ultrasound provides valuable information to the right people at the right time. It’s designed to drive transformation in care delivery and digital health – a dynamic combination that can extend the reach of ultrasound in a remarkable way.”
Related Links:
SMACC
Royal Philips
Read the full article by registering today, it's FREE! 

Register now for FREE to MedImaging.net and get complete access to news and events that shape the world of Radiology. 
- Free digital version edition of Medical Imaging International sent by email on regular basis
- Free print version of Medical Imaging International magazine (available only outside USA and Canada).
- Free and unlimited access to back issues of Medical Imaging International in digital format
- Free Medical Imaging International Newsletter sent every week containing the latest news
- Free breaking news sent via email
- Free access to Events Calendar
- Free access to LinkXpress new product services
- REGISTRATION IS FREE AND EASY!
Sign in: Registered website members
Sign in: Registered magazine subscribers
Latest Ultrasound News
- First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity

- Largest Model Trained On Echocardiography Images Assesses Heart Structure and Function
- Groundbreaking Technology Enables Precise, Automatic Measurement of Peripheral Blood Vessels
- Deep Learning Advances Super-Resolution Ultrasound Imaging
- Novel Ultrasound-Launched Targeted Nanoparticle Eliminates Biofilm and Bacterial Infection
- AI-Guided Ultrasound System Enables Rapid Assessments of DVT
- Focused Ultrasound Technique Gets Quality Assurance Protocol
- AI-Guided Handheld Ultrasound System Helps Capture Diagnostic-Quality Cardiac Images
- Non-Invasive Ultrasound Imaging Device Diagnoses Risk of Chronic Kidney Disease
- Wearable Ultrasound Platform Paves Way for 24/7 Blood Pressure Monitoring On the Wrist
- Diagnostic Ultrasound Enhancing Agent to Improve Image Quality in Pediatric Heart Patients
- AI Detects COVID-19 in Lung Ultrasound Images
- New Ultrasound Technology to Revolutionize Respiratory Disease Diagnoses
- Dynamic Contrast-Enhanced Ultrasound Highly Useful For Interventions
- Ultrasensitive Broadband Transparent Ultrasound Transducer Enhances Medical Diagnosis
- Artificial Intelligence Detects Heart Defects in Newborns from Ultrasound Images
Channels
Radiography
view channel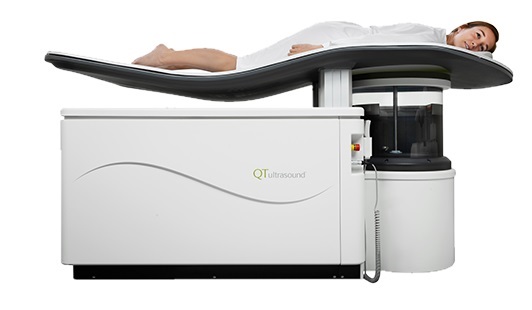
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more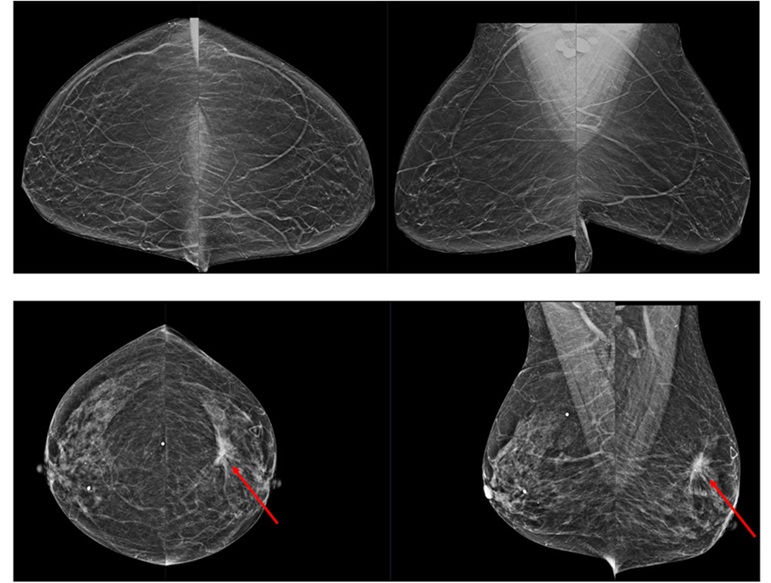
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreMRI
view channel
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read more
World's First Sensor Detects Errors in MRI Scans Using Laser Light and Gas
MRI scanners are daily tools for doctors and healthcare professionals, providing unparalleled 3D imaging of the brain, vital organs, and soft tissues, far surpassing other imaging technologies in quality.... Read more
Diamond Dust Could Offer New Contrast Agent Option for Future MRI Scans
Gadolinium, a heavy metal used for over three decades as a contrast agent in medical imaging, enhances the clarity of MRI scans by highlighting affected areas. Despite its utility, gadolinium not only... Read more.jpg)
Combining MRI with PSA Testing Improves Clinical Outcomes for Prostate Cancer Patients
Prostate cancer is a leading health concern globally, consistently being one of the most common types of cancer among men and a major cause of cancer-related deaths. In the United States, it is the most... Read moreNuclear Medicine
view channel
New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
Imaging inflammation using traditional radiological techniques presents significant challenges, including radiation exposure, poor image quality, high costs, and invasive procedures. Now, new contrast... Read more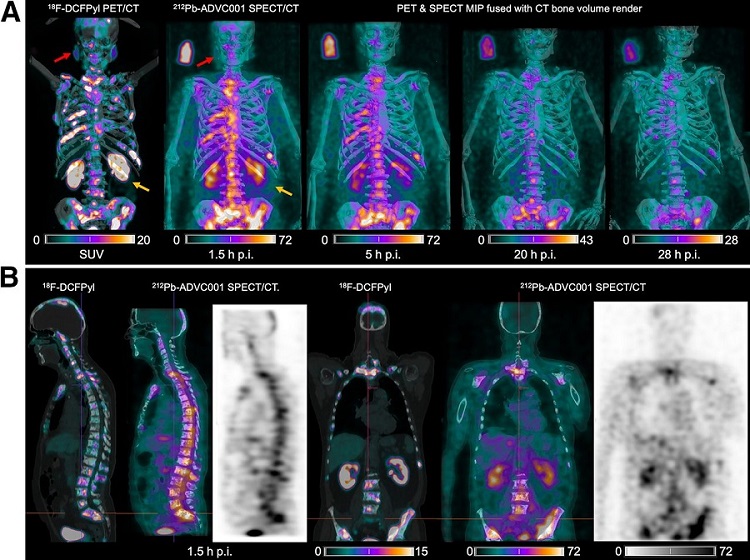
New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
The development of lead-212 (212Pb)-PSMA–based targeted alpha therapy (TAT) is garnering significant interest in treating patients with metastatic castration-resistant prostate cancer. The imaging of 212Pb,... Read moreNew Radiotheranostic System Detects and Treats Ovarian Cancer Noninvasively
Ovarian cancer is the most lethal gynecological cancer, with less than a 30% five-year survival rate for those diagnosed in late stages. Despite surgery and platinum-based chemotherapy being the standard... Read more
AI System Automatically and Reliably Detects Cardiac Amyloidosis Using Scintigraphy Imaging
Cardiac amyloidosis, a condition characterized by the buildup of abnormal protein deposits (amyloids) in the heart muscle, severely affects heart function and can lead to heart failure or death without... Read moreGeneral/Advanced Imaging
view channel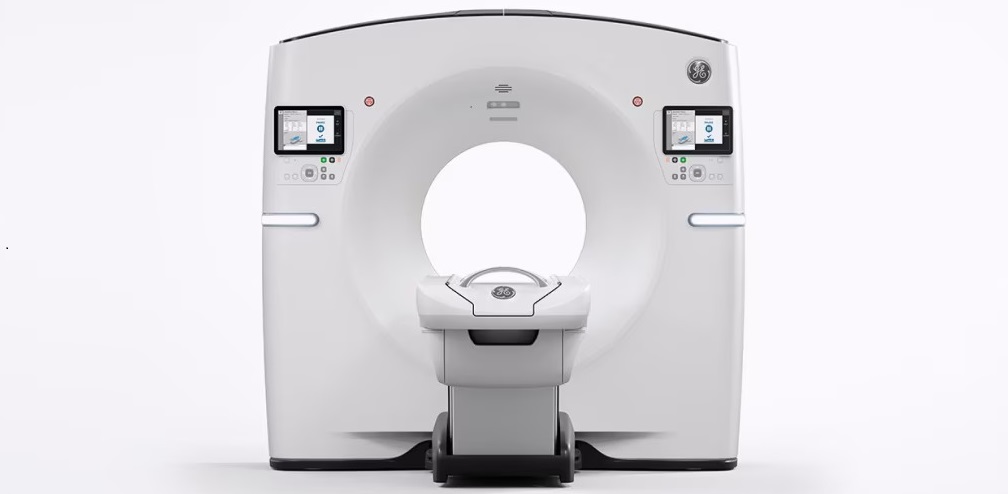
Radiation Therapy Computed Tomography Solution Boosts Imaging Accuracy
One of the most significant challenges in oncology care is disease complexity in terms of the variety of cancer types and the individualized presentation of each patient. This complexity necessitates a... Read more
PET Scans Reveal Hidden Inflammation in Multiple Sclerosis Patients
A key challenge for clinicians treating patients with multiple sclerosis (MS) is that after a certain amount of time, they continue to worsen even though their MRIs show no change. A new study has now... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more