New Tumor-Targeting Agent Images a Wide Range of Cancers
|
By MedImaging International staff writers Posted on 30 Jun 2014 |
Scientists reported that a new type of tumor-targeting agent can search out and target dozens of solid tumors, even illuminating brain cancer stem cells that do not respond to current treatments.
Moreover, years of animal studies and early human clinical trials show that this tumor-targeting, alkylphosphocholine (APC) molecule can deliver two types of “cargoes” right to cancer cells: a radioactive or fluorescent imaging label, or a radioactive medicine that binds and destroys cancer cells.
The study’s findings were reported in the June 13, 2014, issue of the journal Science Translational Medicine. The APC targeting platform is a synthetic molecule that exploits a weakness common to cancers as diverse as breast, lung, brain, and melanoma. These cancer cells lack the enzymes to metabolize phospholipid ethers, a cell membrane component that is easily cleared by normal cells. When administered in an intravenous solution, APC goes throughout the body—even across the blood-brain barrier—and adhere to the membrane of cancer cells. The cancer cells take up the APC and the imaging or treatment medication riding on the molecular platform, and retain it for days to weeks, resulting in direct cancer cell imaging or treatment.
The APC analogs were able to tag 55 of 57 different cancers. This large study had multiple stages, including testing in cancer cell lines, in rodents and rodents infected with human and rodent cancers, and in human patients with different cancers such as colorectal, breast, lung, and glioblastoma.
“I was a skeptic; it’s almost too good to be true,” said co-lead author Dr. John S. Kuo, associate professor of neurosurgery and director of the comprehensive brain tumor program at the University of Wisconsin (UW) School of Medicine and Public Health (Madison, USA). “It is a very broad cancer-targeting agent in terms of the many different cancers that tested positive. The APC analogs even sometimes revealed other sites of cancer in patients that were small, asymptomatic and previously undetected by physicians.”
Dr. Kuo specializes in the treatment of brain tumors, and also leads the UWCCC CNS Tumors group that are conducting many clinical trials for glioma, a brain cancer that is incurable because current treatments leave behind cancer stem cells that can seed and regrow the cancer. He noted that it was encouraging that the APC analogs also captured cancer stem cells and will also likely target them for additional treatment. “It’s also potentially superior to current imaging methods because the standard clinical MR or PET imaging may give false-positive results due to surgical scars, post-treatment effects, inflammation, or even infection, making it difficult to know if the cancer has truly returned,” he stated.
Dr. Kuo noted that the fluorescent intraoperative APC imaging might help make cancer surgeries more effective and safer; any cancer cells that cannot be safely removed can be targeted afterwards with radioactive APC therapy. In addition, APC imaging might avoid the false-positive findings of current imaging, so cancer patients can stay on effective therapies and likely avoid the risks and costs of “second look” surgeries.
The large multidisciplinary study team was also led by co-lead author Dr. Jamey P. Weichert, associate professor of radiology, who cofounded and serves as chief scientific officer of Cellectar Biosciences, Inc., a Madison-based company developing the APC analog platform for cancer imaging and therapy.
Related Links:
University of Wisconsin School of Medicine and Public Health
Moreover, years of animal studies and early human clinical trials show that this tumor-targeting, alkylphosphocholine (APC) molecule can deliver two types of “cargoes” right to cancer cells: a radioactive or fluorescent imaging label, or a radioactive medicine that binds and destroys cancer cells.
The study’s findings were reported in the June 13, 2014, issue of the journal Science Translational Medicine. The APC targeting platform is a synthetic molecule that exploits a weakness common to cancers as diverse as breast, lung, brain, and melanoma. These cancer cells lack the enzymes to metabolize phospholipid ethers, a cell membrane component that is easily cleared by normal cells. When administered in an intravenous solution, APC goes throughout the body—even across the blood-brain barrier—and adhere to the membrane of cancer cells. The cancer cells take up the APC and the imaging or treatment medication riding on the molecular platform, and retain it for days to weeks, resulting in direct cancer cell imaging or treatment.
The APC analogs were able to tag 55 of 57 different cancers. This large study had multiple stages, including testing in cancer cell lines, in rodents and rodents infected with human and rodent cancers, and in human patients with different cancers such as colorectal, breast, lung, and glioblastoma.
“I was a skeptic; it’s almost too good to be true,” said co-lead author Dr. John S. Kuo, associate professor of neurosurgery and director of the comprehensive brain tumor program at the University of Wisconsin (UW) School of Medicine and Public Health (Madison, USA). “It is a very broad cancer-targeting agent in terms of the many different cancers that tested positive. The APC analogs even sometimes revealed other sites of cancer in patients that were small, asymptomatic and previously undetected by physicians.”
Dr. Kuo specializes in the treatment of brain tumors, and also leads the UWCCC CNS Tumors group that are conducting many clinical trials for glioma, a brain cancer that is incurable because current treatments leave behind cancer stem cells that can seed and regrow the cancer. He noted that it was encouraging that the APC analogs also captured cancer stem cells and will also likely target them for additional treatment. “It’s also potentially superior to current imaging methods because the standard clinical MR or PET imaging may give false-positive results due to surgical scars, post-treatment effects, inflammation, or even infection, making it difficult to know if the cancer has truly returned,” he stated.
Dr. Kuo noted that the fluorescent intraoperative APC imaging might help make cancer surgeries more effective and safer; any cancer cells that cannot be safely removed can be targeted afterwards with radioactive APC therapy. In addition, APC imaging might avoid the false-positive findings of current imaging, so cancer patients can stay on effective therapies and likely avoid the risks and costs of “second look” surgeries.
The large multidisciplinary study team was also led by co-lead author Dr. Jamey P. Weichert, associate professor of radiology, who cofounded and serves as chief scientific officer of Cellectar Biosciences, Inc., a Madison-based company developing the APC analog platform for cancer imaging and therapy.
Related Links:
University of Wisconsin School of Medicine and Public Health
Latest Nuclear Medicine News
- New PET Biomarker Predicts Success of Immune Checkpoint Blockade Therapy
- New PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
- New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
- New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
- New Radiotheranostic System Detects and Treats Ovarian Cancer Noninvasively
- AI System Automatically and Reliably Detects Cardiac Amyloidosis Using Scintigraphy Imaging
- Early 30-Minute Dynamic FDG-PET Acquisition Could Halve Lung Scan Times
- New Method for Triggering and Imaging Seizures to Help Guide Epilepsy Surgery
- Radioguided Surgery Accurately Detects and Removes Metastatic Lymph Nodes in Prostate Cancer Patients
- New PET Tracer Detects Inflammatory Arthritis Before Symptoms Appear
- Novel PET Tracer Enhances Lesion Detection in Medullary Thyroid Cancer
- Targeted Therapy Delivers Radiation Directly To Cells in Hard-To-Treat Cancers
- New PET Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Algorithm Predicts Prostate Cancer Recurrence in Patients Treated by Radiation Therapy
- Novel PET Imaging Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Ultrafast Laser Technology to Improve Cancer Treatment
Channels
Radiography
view channel
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more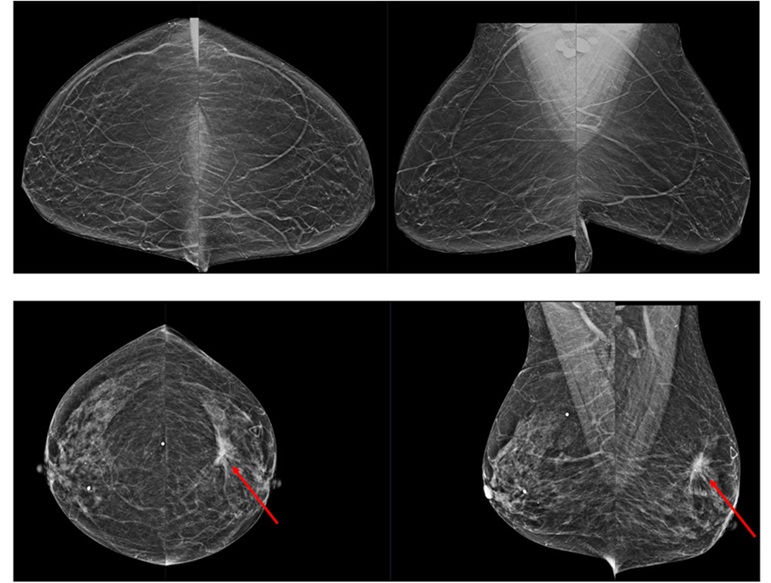
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreMRI
view channel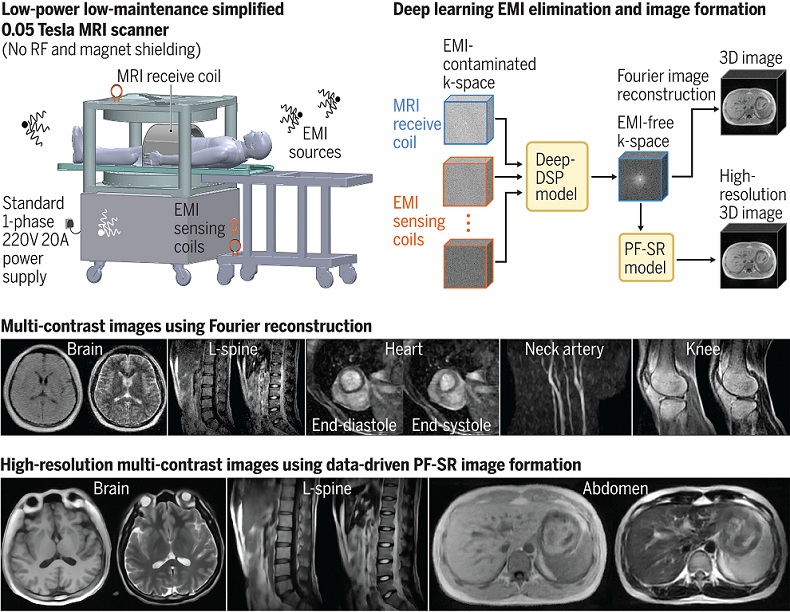
Low-Cost Whole-Body MRI Device Combined with AI Generates High-Quality Results
Magnetic Resonance Imaging (MRI) has significantly transformed healthcare, providing a noninvasive, radiation-free method for detailed imaging. It is especially promising for the future of medical diagnosis... Read more
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read moreUltrasound
view channel.jpg)
Diagnostic System Automatically Analyzes TTE Images to Identify Congenital Heart Disease
Congenital heart disease (CHD) is one of the most prevalent congenital anomalies worldwide, presenting substantial health and financial challenges for affected patients. Early detection and treatment of... Read more
Super-Resolution Imaging Technique Could Improve Evaluation of Cardiac Conditions
The heart depends on efficient blood circulation to pump blood throughout the body, delivering oxygen to tissues and removing carbon dioxide and waste. Yet, when heart vessels are damaged, it can disrupt... Read more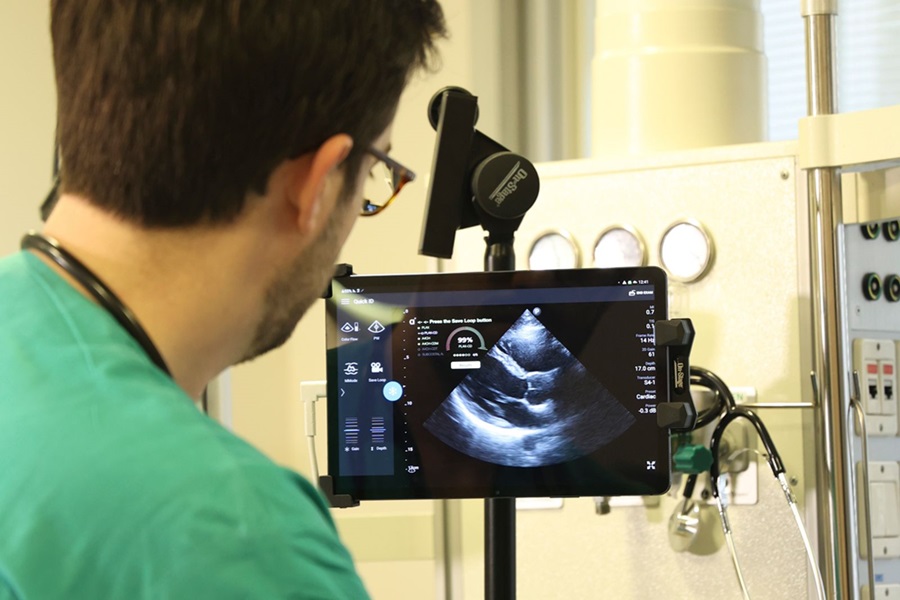
First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity
Ultrasound scans are essential for identifying and diagnosing various medical conditions, but often, patients must wait weeks or months for results due to a shortage of qualified medical professionals... Read moreGeneral/Advanced Imaging
view channelBone Density Test Uses Existing CT Images to Predict Fractures
Osteoporotic fractures are not only devastating and deadly, especially hip fractures, but also impose significant costs. They rank among the top chronic diseases in terms of disability-adjusted life years... Read more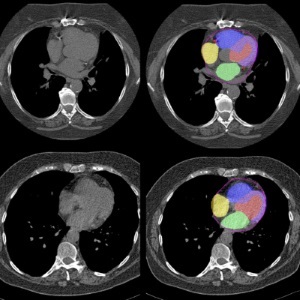
AI Predicts Cardiac Risk and Mortality from Routine Chest CT Scans
Heart disease remains the leading cause of death and is largely preventable, yet many individuals are unaware of their risk until it becomes severe. Early detection through screening can reveal heart issues,... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more