PET Imaging Used to Assess Effectiveness of Menkes Disease Treatments
|
By MedImaging International staff writers Posted on 15 Apr 2014 |
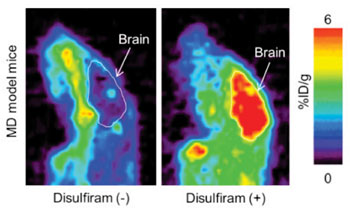
Image: Sagittal section of brain PET image at four hours after 64CuCl2 injection with disulfiram or D-penicillamine in MD model mice (Photo courtesy of the RIKEN Center for Life Science Technologies).
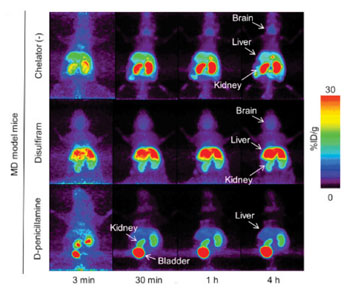
Image: Coronal section of whole-body PET image at four hours after 64CuCl2 injection with disulfiram or D-penicillamine in MD model mice (Photo courtesy of the RIKEN Center for Life Science Technologies).
Japanese scientists are using positron emission tomography (PET) imaging to visualize the distribution of copper in the body using lab mice. Copper distribution is deregulated in a genetic disorder called Menkes disease (MD). This study provides the groundwork for PET imaging studies on human MD patients to identify new therapy approaches.
Although rare, Menkes disease, is a formidable genetic disorder. Most affected infants die within the first few years of life. The disease is caused by an inborn defect in the body’s ability to absorb copper. The standard treatment today for the 1 in 100,000 babies affected by the disorder is to inject copper, but this therapy has limited efficacy. Eventually the treatment becomes ineffective, leading to neurodeneration, and the copper accumulates in the kidneys, sometimes leading to renal failure.
The resulting treatments have been found to enhance the accumulation of injected copper in the brain while preventing its accumulation in the kidney. Recently, disulfiram, a medication developed to treat alcoholism, has been proposed as a therapy for MD, since one of its actions is to enhance this copper accumulation in the brain.
Now, in a new study published April 1, 2014, in the Journal of Nuclear Medicine, scientists from the RIKEN Center for Life Science Technologies (Kobe, Japan), working with pediatricians from Osaka City University (Japan) and Teikyo University (Tokyo, Japan) employed positron emission tomography imaging (PET) imaging to show that a combination of copper injections and disulfiram or D-penicillamine allows a greater movement of copper to the brain, where it is needed, without accruing in the kidneys.
In the study, the researchers used Menkes disease model mice, which have an inborn defect in copper metabolism, and injected copper-64, a radioactive isotope of copper, into the mice. They then used PET scanning, a noninvasive procedure, to visualize how the copper traveled throughout the body. They compared mice injected with copper alone to mice injected with copper in addition to one of two other drugs, disulfiram, or D-penicillamine, and the copper distribution throughout the body was observed for a four-hour time period.
The findings revealed that the mice administered the copper combined with disulfiram had a comparatively high concentration of copper in the brain without a substantial increase in the kidneys. Remarkably, it revealed that the amount of copper going to the brain in mice treated with disulfiram was in reality higher than in those treated with copper by itself, suggesting that the agent has an effect on the passage of copper through the blood-brain barrier.
According to Dr. Satoshi Nozaki, one of the co-authors, “This study demonstrates that PET imaging can be a useful tool for evaluating new treatments for Menkes disease.” Planning for the future, he said, “Based on this study, we are planning to conduct clinical PET studies of patient with Menkes disease.”
Related Links:
RIKEN Center for Life Science Technologies
Osaka City University
Teikyo University
Although rare, Menkes disease, is a formidable genetic disorder. Most affected infants die within the first few years of life. The disease is caused by an inborn defect in the body’s ability to absorb copper. The standard treatment today for the 1 in 100,000 babies affected by the disorder is to inject copper, but this therapy has limited efficacy. Eventually the treatment becomes ineffective, leading to neurodeneration, and the copper accumulates in the kidneys, sometimes leading to renal failure.
The resulting treatments have been found to enhance the accumulation of injected copper in the brain while preventing its accumulation in the kidney. Recently, disulfiram, a medication developed to treat alcoholism, has been proposed as a therapy for MD, since one of its actions is to enhance this copper accumulation in the brain.
Now, in a new study published April 1, 2014, in the Journal of Nuclear Medicine, scientists from the RIKEN Center for Life Science Technologies (Kobe, Japan), working with pediatricians from Osaka City University (Japan) and Teikyo University (Tokyo, Japan) employed positron emission tomography imaging (PET) imaging to show that a combination of copper injections and disulfiram or D-penicillamine allows a greater movement of copper to the brain, where it is needed, without accruing in the kidneys.
In the study, the researchers used Menkes disease model mice, which have an inborn defect in copper metabolism, and injected copper-64, a radioactive isotope of copper, into the mice. They then used PET scanning, a noninvasive procedure, to visualize how the copper traveled throughout the body. They compared mice injected with copper alone to mice injected with copper in addition to one of two other drugs, disulfiram, or D-penicillamine, and the copper distribution throughout the body was observed for a four-hour time period.
The findings revealed that the mice administered the copper combined with disulfiram had a comparatively high concentration of copper in the brain without a substantial increase in the kidneys. Remarkably, it revealed that the amount of copper going to the brain in mice treated with disulfiram was in reality higher than in those treated with copper by itself, suggesting that the agent has an effect on the passage of copper through the blood-brain barrier.
According to Dr. Satoshi Nozaki, one of the co-authors, “This study demonstrates that PET imaging can be a useful tool for evaluating new treatments for Menkes disease.” Planning for the future, he said, “Based on this study, we are planning to conduct clinical PET studies of patient with Menkes disease.”
Related Links:
RIKEN Center for Life Science Technologies
Osaka City University
Teikyo University
Latest Nuclear Medicine News
- New PET Biomarker Predicts Success of Immune Checkpoint Blockade Therapy
- New PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
- New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
- New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
- New Radiotheranostic System Detects and Treats Ovarian Cancer Noninvasively
- AI System Automatically and Reliably Detects Cardiac Amyloidosis Using Scintigraphy Imaging
- Early 30-Minute Dynamic FDG-PET Acquisition Could Halve Lung Scan Times
- New Method for Triggering and Imaging Seizures to Help Guide Epilepsy Surgery
- Radioguided Surgery Accurately Detects and Removes Metastatic Lymph Nodes in Prostate Cancer Patients
- New PET Tracer Detects Inflammatory Arthritis Before Symptoms Appear
- Novel PET Tracer Enhances Lesion Detection in Medullary Thyroid Cancer
- Targeted Therapy Delivers Radiation Directly To Cells in Hard-To-Treat Cancers
- New PET Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Algorithm Predicts Prostate Cancer Recurrence in Patients Treated by Radiation Therapy
- Novel PET Imaging Tracer Noninvasively Identifies Cancer Gene Mutation for More Precise Diagnosis
- Ultrafast Laser Technology to Improve Cancer Treatment
Channels
Radiography
view channel
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreMRI
view channel
Low-Cost Whole-Body MRI Device Combined with AI Generates High-Quality Results
Magnetic Resonance Imaging (MRI) has significantly transformed healthcare, providing a noninvasive, radiation-free method for detailed imaging. It is especially promising for the future of medical diagnosis... Read more
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read moreUltrasound
view channel.jpg)
Diagnostic System Automatically Analyzes TTE Images to Identify Congenital Heart Disease
Congenital heart disease (CHD) is one of the most prevalent congenital anomalies worldwide, presenting substantial health and financial challenges for affected patients. Early detection and treatment of... Read more
Super-Resolution Imaging Technique Could Improve Evaluation of Cardiac Conditions
The heart depends on efficient blood circulation to pump blood throughout the body, delivering oxygen to tissues and removing carbon dioxide and waste. Yet, when heart vessels are damaged, it can disrupt... Read more
First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity
Ultrasound scans are essential for identifying and diagnosing various medical conditions, but often, patients must wait weeks or months for results due to a shortage of qualified medical professionals... Read moreGeneral/Advanced Imaging
view channelBone Density Test Uses Existing CT Images to Predict Fractures
Osteoporotic fractures are not only devastating and deadly, especially hip fractures, but also impose significant costs. They rank among the top chronic diseases in terms of disability-adjusted life years... Read more
AI Predicts Cardiac Risk and Mortality from Routine Chest CT Scans
Heart disease remains the leading cause of death and is largely preventable, yet many individuals are unaware of their risk until it becomes severe. Early detection through screening can reveal heart issues,... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more