Pacing Lead for Full-Body MRI Scans Approved for Use in Patients with a Slow Heartbeat
|
By MedImaging International staff writers Posted on 21 Oct 2014 |
A new magnetic resonance imaging (MRI) pacing lead is approved in the United States for MRI scans positioned on any region of the body when used with a Medtronic dual-chamber MR-conditional pacemaker for patients with a slow heartbeat.
Medtronic, Inc. (Minneapolis, MN, USA) announced the US Food and Drug Administration (FDA) approval of its CapSureFix Novus MRI SureScan 5076 lead, previously approved for use with Medtronic’s non-MR-conditional pacemakers, has been well-received because of its handling and effectiveness. Since its introduction more than 10 years ago, physicians have implanted more than three million 5076 leads in patients.
With the new FDA approval, two 5076 MRI leads can now be paired with dual-chamber Medtronic Advisa MRI or Revo MRI SureScan pacemakers, allowing patients with these complete SureScan pacing systems to undergo full-body MRI scans. Medtronic 5076 MRI lead lengths approved for these scans range in length from 35–85 cm. Patients who previously had two 5076 leads implanted with non-MRI pacemakers will have the option to receive MRIs if MR-conditional SureScan pacemakers are implanted when replacement devices are needed.
“The 5076 lead has proven to be one of the most reliable pacing leads for more than a decade, and due to extensive testing, now also can undergo MRI scans,” said Brian Urke, vice president and general manager of the bradycardia business at Medtronic. “This is especially important for patients who need MRIs and received 5076 leads at the time of their initial implants; although they may not have received MR-conditional pacemakers initially, they now have the option to get a pacemaker approved for MRI when they require a device change-out, making the entire system MR-conditional.”
Since its first MR-conditional pacing system was approved in Europe in 2008, Medtronic has continued to design and test its products for safe use during MRI scanning. Recent developments in computer modeling have allowed Medtronic to evaluate the 5076 lead across more than two million scanning situations. Furthermore, Medtronic conducted the 5076 MRI Clinical Study, global, multicenter research to evaluate the safety and effectiveness of the 5076 lead in the MRI environment.
Worldwide, it is estimated that up to 75% of patients with implanted cardiac devices are expected to need an MRI scan during the lifetime of their devices. MRI is the standard of care in soft tissue imaging, providing data not seen with X-ray, ultrasound, or computed tomography (CT) scan. MRI is therefore crucial for the early detection and treatment of many diseases. Until recently, patients with implanted pacemakers were refused access to MRI procedures because the interaction can be injurious.
The 5076 MRI lead is the latest addition to a range of Medtronic devices that are approved for MRI access. These include the Medtronic SureScan pacing systems, the SureScan neurostimulation systems for the management of chronic pain, and the SynchroMed II programmable drug infusion system, which are available worldwide. Moreover, the Evera MRI SureScan implantable cardioverter-defibrillator (ICD) system is investigational in the United States.
In collaboration with leading clinicians, researchers, and scientists worldwide, Medtronic offers a wide range of novel medical technology for the interventional and surgical treatment of cardiovascular disease and cardiac arrhythmias.
Related Links:
Medtronic
Medtronic, Inc. (Minneapolis, MN, USA) announced the US Food and Drug Administration (FDA) approval of its CapSureFix Novus MRI SureScan 5076 lead, previously approved for use with Medtronic’s non-MR-conditional pacemakers, has been well-received because of its handling and effectiveness. Since its introduction more than 10 years ago, physicians have implanted more than three million 5076 leads in patients.
With the new FDA approval, two 5076 MRI leads can now be paired with dual-chamber Medtronic Advisa MRI or Revo MRI SureScan pacemakers, allowing patients with these complete SureScan pacing systems to undergo full-body MRI scans. Medtronic 5076 MRI lead lengths approved for these scans range in length from 35–85 cm. Patients who previously had two 5076 leads implanted with non-MRI pacemakers will have the option to receive MRIs if MR-conditional SureScan pacemakers are implanted when replacement devices are needed.
“The 5076 lead has proven to be one of the most reliable pacing leads for more than a decade, and due to extensive testing, now also can undergo MRI scans,” said Brian Urke, vice president and general manager of the bradycardia business at Medtronic. “This is especially important for patients who need MRIs and received 5076 leads at the time of their initial implants; although they may not have received MR-conditional pacemakers initially, they now have the option to get a pacemaker approved for MRI when they require a device change-out, making the entire system MR-conditional.”
Since its first MR-conditional pacing system was approved in Europe in 2008, Medtronic has continued to design and test its products for safe use during MRI scanning. Recent developments in computer modeling have allowed Medtronic to evaluate the 5076 lead across more than two million scanning situations. Furthermore, Medtronic conducted the 5076 MRI Clinical Study, global, multicenter research to evaluate the safety and effectiveness of the 5076 lead in the MRI environment.
Worldwide, it is estimated that up to 75% of patients with implanted cardiac devices are expected to need an MRI scan during the lifetime of their devices. MRI is the standard of care in soft tissue imaging, providing data not seen with X-ray, ultrasound, or computed tomography (CT) scan. MRI is therefore crucial for the early detection and treatment of many diseases. Until recently, patients with implanted pacemakers were refused access to MRI procedures because the interaction can be injurious.
The 5076 MRI lead is the latest addition to a range of Medtronic devices that are approved for MRI access. These include the Medtronic SureScan pacing systems, the SureScan neurostimulation systems for the management of chronic pain, and the SynchroMed II programmable drug infusion system, which are available worldwide. Moreover, the Evera MRI SureScan implantable cardioverter-defibrillator (ICD) system is investigational in the United States.
In collaboration with leading clinicians, researchers, and scientists worldwide, Medtronic offers a wide range of novel medical technology for the interventional and surgical treatment of cardiovascular disease and cardiac arrhythmias.
Related Links:
Medtronic
Latest MRI News
- Cardiac MRI Measure Improves Risk Prediction in Tricuspid Regurgitation
- AI System Improves Accuracy of Cardiac MRI Interpretation
- Deep Learning Model Predicts Alzheimer’s Disease Outcomes from Baseline MRI
- Blood-Brain Barrier Imaging Adds Risk Insight to Standard Stroke MRI
- AI Body Composition MRI Analysis Predicts Cardiometabolic Disease Risk
- AI MRI Tool Quantifies Muscle Fat to Assess Cardiometabolic Risk
- Advanced MRI Visualizes CSF Motion Changes After Mild Traumatic Brain Injury
- MRI Tool Enables Long-Term Tracking of Transplanted Cardiac Cells
- MRI-Based AI Tool Supports Differentiation of Parkinsonian Syndromes
- MRI-Derived Biomarker Improves Risk Stratification in Glioblastoma
- Combined Imaging Approach Identifies Cause of Heart Attack without Coronary Blockage
- Advanced MRI System Detects Impaired Cardiac Oxygen Use in Minutes
- AI-Enhanced MRI Improves Image Quality in Arrhythmia Patients
- Ultra-Detailed Brain Atlas Enhances Early Detection of Neurological Disorders
- Study Finds Advanced Imaging Significantly Reduces Unnecessary Prostate Biopsies
- New Material Boosts MRI Image Quality
Channels
Radiography
view channel
Simple Chest X-Ray Measure Predicts Survival After Lung Cancer Surgery
Obstructive ventilatory disorder, marked by airflow limitation that reduces breathing efficiency, increases postoperative risk in patients with lung cancer. Although surgery offers the best chance of cure,... Read more
AI Detection Tool Improves Identification of Lobular Breast Cancer
Breast cancer screening seeks early detection, yet some subtypes remain difficult to visualize on mammography, risking delayed diagnosis. On average, 1 in 20 women worldwide will develop breast cancer,... Read moreUltrasound
view channelAI Robotic Ultrasound System Automates Echocardiography and Improves Consistency
Echocardiography, an ultrasound examination of the heart, is central to diagnosing and managing cardiovascular disease. Many services struggle with limited availability of skilled sonographers, variable... Read more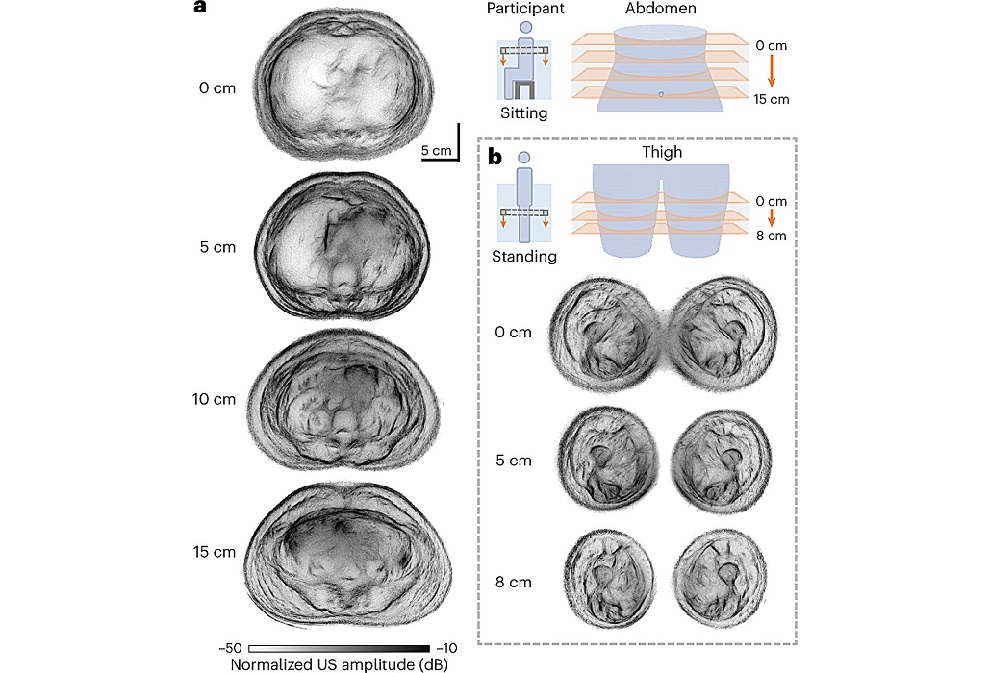
Whole Cross-Section Ultrasound System Enables Operator-Independent Imaging
Conventional ultrasound is central to bedside imaging but is limited by a narrow field of view and operator variability. Comprehensive cross-sectional assessment typically requires computed tomography... Read moreNuclear Medicine
view channelMR-Guided Cardiac Mapping System Enables Radiation-Free Procedures
Cardiac electrophysiology procedures are typically guided by X-ray fluoroscopy, which limits soft-tissue visualization and exposes patients and clinical staff to ionizing radiation. Real-time mapping that... Read more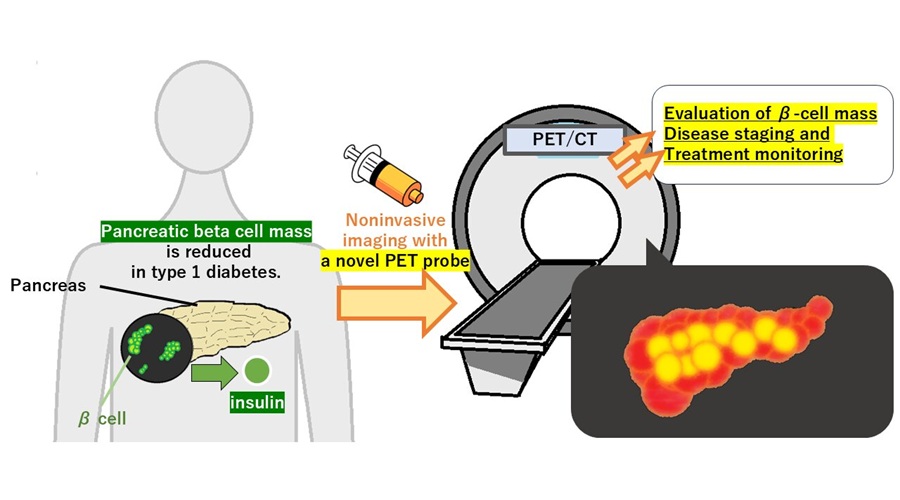
PET Tracer Enables Noninvasive Measurement of Beta Cell Mass
Type 1 diabetes is an autoimmune disease in which the immune system destroys insulin-producing pancreatic beta cells. Loss of these cells destabilizes glucose control and drives complications.... Read more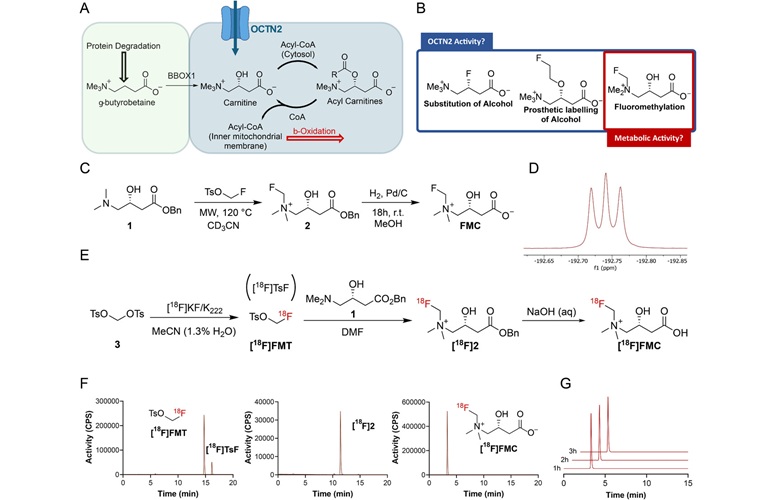
New Imaging Tool Sheds Light on Tumor Fat Metabolism
Rapidly growing tumors reprogram metabolism to meet high energy demands. While many cancers preferentially consume glucose, lipid utilization by malignant cells is difficult to measure in living subjects.... Read more
Radiopharmaceutical Molecule Marker to Improve Choice of Bladder Cancer Therapies
Targeted cancer therapies only work when tumor cells express the specific molecular structures they are designed to attack. In urothelial carcinoma, a common form of bladder cancer, the cell surface protein... Read moreGeneral/Advanced Imaging
view channelMultimodal AI Tool Combines CT and Health Records to Predict Heart Risk
Cardiovascular disease is a leading cause of death and an underrecognized risk for people treated for breast cancer. Cardiac complications can affect survival and quality of life. Clinicians need tools... Read more
AI Tool Automates Radiotherapy Planning for Cervical and Prostate Cancer
Cervical cancer causes most of its global mortality in low- and middle-income countries, where radiotherapy capacity and specialist staff are limited. Treatment planning is labor-intensive and can delay... Read moreImaging IT
view channel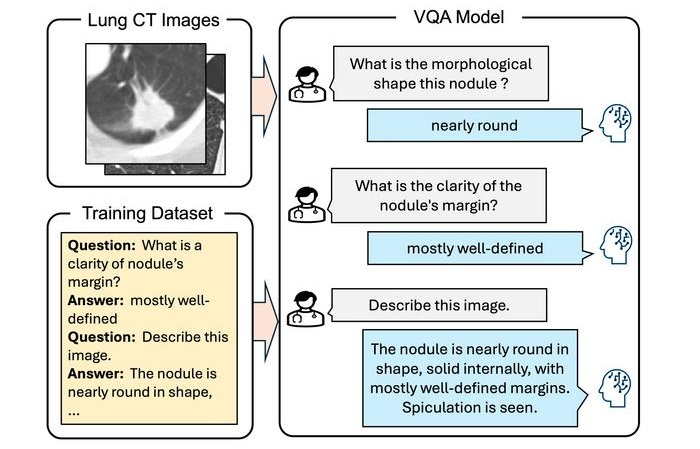
Interactive AI Tool Supports Explainable Lung Nodule Assessment
Lung cancer is a leading cause of cancer mortality, and timely characterization of pulmonary nodules on chest computed tomography (CT) is essential for directing care. Interpreting nodule morphology demands... Read more
Breast Imaging Software Enhances Visualization and Tissue Characterization in Challenging Cases
Breast imaging can be particularly challenging in cases involving small breasts or implants, where image reconstruction and tissue characterization may be limited. Clinicians also need reproducible analysis... Read more
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel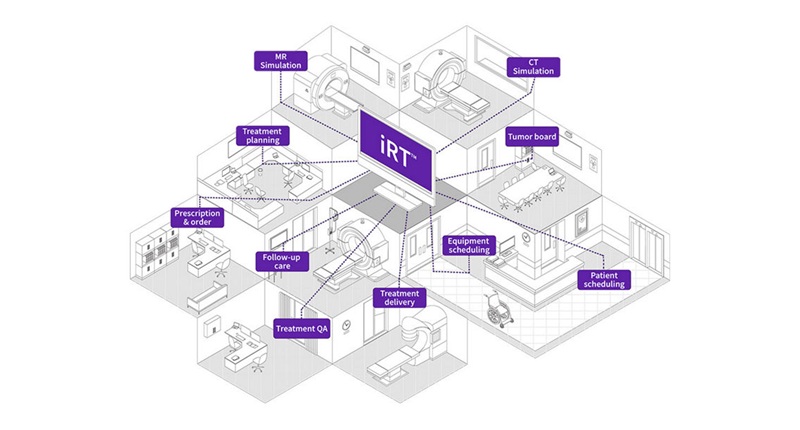
GE HealthCare Highlights AI-Supported Radiation Therapy Tools at ESTRO 2026
At the European Society for Radiotherapy and Oncology (ESTRO) 2026 Congress in Stockholm, GE HealthCare is highlighting Intelligent Radiation Therapy (iRT), MIM Software innovations, and BK Medical surgical... Read more