Standard Phantom Developed for Breast Cancer MRIs
|
By MedImaging International staff writers Posted on 03 Apr 2016 |

Image: NIST engineer Katy Keenan holds the new phantom (Photo courtesy of NIST).
The US National Institute of Standards and Technology (NIST; Gaithersburg, MD, USA) has developed a standardized phantom to help test the performance of magnetic resonance imaging (MRI) devices used to identify and monitor breast cancer.
The NIST phantom will help standardize and ensure quality control of breast tissue MRIs when comparing images within and between medical research studies, comparing MRI scanner performance, and the training of operators. The phantom is composed of a flexible, soft 15 cm × 12.5 cm silicone shell, with internal components made of rigid polycarbonate to ensure regular geometry. The phantom also supports quantitative MRI, which is increasingly being used for breast cancer diagnosis, staging, and treatment monitoring.
The phantom has two basic types of internal arrangements, which can be paired for MRI scans. One unit, designed for conventional MRI scans, is based on the magnetic properties of hydrogen atoms. It contains four layers of small plastic spheres filled with solutions that mimic tissue, including corn syrup in water to represent fibroglandular tissue, and grapeseed oil to represent fat; the spheres can be customized to meet special clinical needs. Potential options include solutions that would mimic either benign cysts or calcium microcalcifications, which can signal cancer.
The second phantom unit is designed for the newer technique of diffusion MRI, which measures the motion of water molecules within breast tissue. The second unit contains plastic vials filled with varying concentrations of a polyvinylpyrrolidone (PVP), a nontoxic polymer which is tuned to mimic the differing diffusion properties of malignant and benign tumors. Both units have a modular design to help fit them into different MRI systems. The development process of the new phantom was published on March 6, 2016, in JMRI.
“The biggest design challenge was to create a lifelike mimic of both fat and fibroglandular tissue. The design success led to industry’s rapid commercialization of the phantom, currently used by five research groups,” said lead author and NIST project leader Katy Keenan, PhD. “I believe the NIST phantom can have a big impact, especially because we are seeing more use of quantitative MRI for breast cancer. It will be used by both researchers developing new techniques and by radiologists using techniques in the clinic.”
Related Links:
US National Institute of Standards and Technology
The NIST phantom will help standardize and ensure quality control of breast tissue MRIs when comparing images within and between medical research studies, comparing MRI scanner performance, and the training of operators. The phantom is composed of a flexible, soft 15 cm × 12.5 cm silicone shell, with internal components made of rigid polycarbonate to ensure regular geometry. The phantom also supports quantitative MRI, which is increasingly being used for breast cancer diagnosis, staging, and treatment monitoring.
The phantom has two basic types of internal arrangements, which can be paired for MRI scans. One unit, designed for conventional MRI scans, is based on the magnetic properties of hydrogen atoms. It contains four layers of small plastic spheres filled with solutions that mimic tissue, including corn syrup in water to represent fibroglandular tissue, and grapeseed oil to represent fat; the spheres can be customized to meet special clinical needs. Potential options include solutions that would mimic either benign cysts or calcium microcalcifications, which can signal cancer.
The second phantom unit is designed for the newer technique of diffusion MRI, which measures the motion of water molecules within breast tissue. The second unit contains plastic vials filled with varying concentrations of a polyvinylpyrrolidone (PVP), a nontoxic polymer which is tuned to mimic the differing diffusion properties of malignant and benign tumors. Both units have a modular design to help fit them into different MRI systems. The development process of the new phantom was published on March 6, 2016, in JMRI.
“The biggest design challenge was to create a lifelike mimic of both fat and fibroglandular tissue. The design success led to industry’s rapid commercialization of the phantom, currently used by five research groups,” said lead author and NIST project leader Katy Keenan, PhD. “I believe the NIST phantom can have a big impact, especially because we are seeing more use of quantitative MRI for breast cancer. It will be used by both researchers developing new techniques and by radiologists using techniques in the clinic.”
Related Links:
US National Institute of Standards and Technology
Latest MRI News
- Low-Cost Whole-Body MRI Device Combined with AI Generates High-Quality Results
- World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
- World's First Sensor Detects Errors in MRI Scans Using Laser Light and Gas
- Diamond Dust Could Offer New Contrast Agent Option for Future MRI Scans
- Combining MRI with PSA Testing Improves Clinical Outcomes for Prostate Cancer Patients
- PET/MRI Improves Diagnostic Accuracy for Prostate Cancer Patients
- Next Generation MR-Guided Focused Ultrasound Ushers In Future of Incisionless Neurosurgery
- Two-Part MRI Scan Detects Prostate Cancer More Quickly without Compromising Diagnostic Quality
- World’s Most Powerful MRI Machine Images Living Brain with Unrivaled Clarity
- New Whole-Body Imaging Technology Makes It Possible to View Inflammation on MRI Scan
- Combining Prostate MRI with Blood Test Can Avoid Unnecessary Prostate Biopsies
- New Treatment Combines MRI and Ultrasound to Control Prostate Cancer without Serious Side Effects
- MRI Improves Diagnosis and Treatment of Prostate Cancer
- Combined PET-MRI Scan Improves Treatment for Early Breast Cancer Patients
- 4D MRI Could Improve Clinical Assessment of Heart Blood Flow Abnormalities
- MRI-Guided Focused Ultrasound Therapy Shows Promise in Treating Prostate Cancer
Channels
Radiography
view channel
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more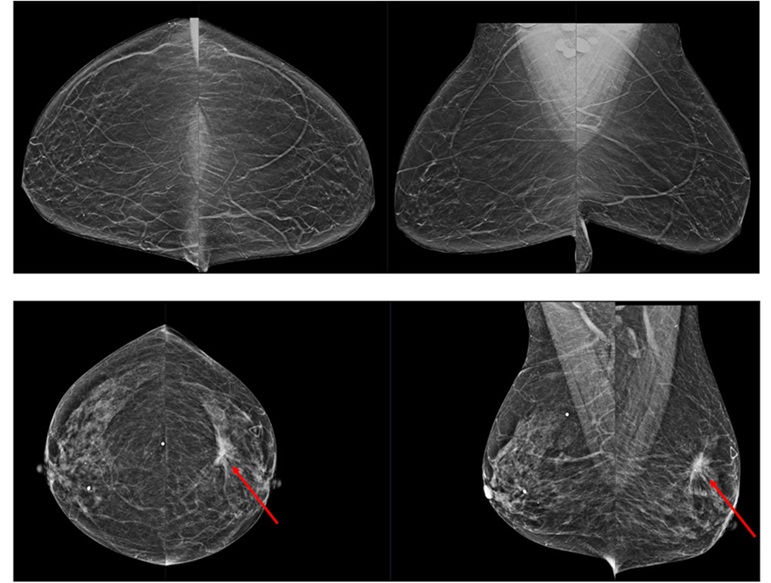
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreUltrasound
view channel.jpg)
Diagnostic System Automatically Analyzes TTE Images to Identify Congenital Heart Disease
Congenital heart disease (CHD) is one of the most prevalent congenital anomalies worldwide, presenting substantial health and financial challenges for affected patients. Early detection and treatment of... Read more
Super-Resolution Imaging Technique Could Improve Evaluation of Cardiac Conditions
The heart depends on efficient blood circulation to pump blood throughout the body, delivering oxygen to tissues and removing carbon dioxide and waste. Yet, when heart vessels are damaged, it can disrupt... Read more
First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity
Ultrasound scans are essential for identifying and diagnosing various medical conditions, but often, patients must wait weeks or months for results due to a shortage of qualified medical professionals... Read moreNuclear Medicine
view channel
New PET Biomarker Predicts Success of Immune Checkpoint Blockade Therapy
Immunotherapies, such as immune checkpoint blockade (ICB), have shown promising clinical results in treating melanoma, non-small cell lung cancer, and other tumor types. However, the effectiveness of these... Read moreNew PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
Clear cell renal cell cancer (ccRCC) represents 70-80% of renal cell carcinoma cases. While localized disease can be effectively treated with surgery and ablative therapies, one-third of patients either... Read more
New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
Imaging inflammation using traditional radiological techniques presents significant challenges, including radiation exposure, poor image quality, high costs, and invasive procedures. Now, new contrast... Read more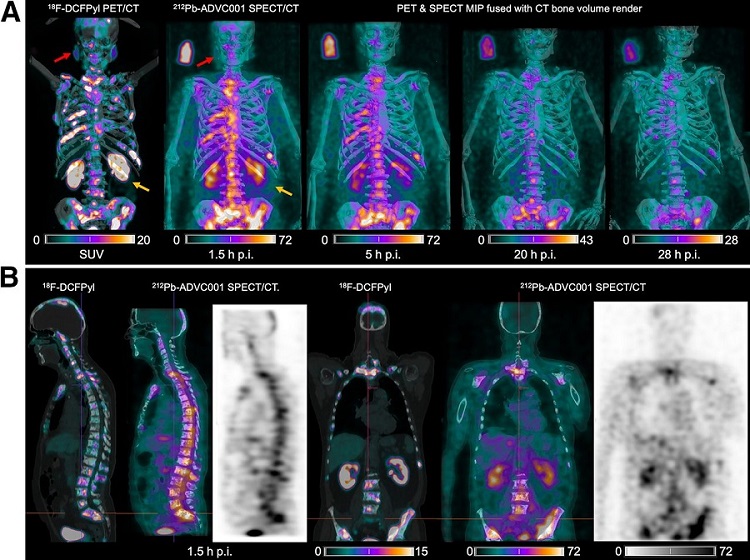
New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
The development of lead-212 (212Pb)-PSMA–based targeted alpha therapy (TAT) is garnering significant interest in treating patients with metastatic castration-resistant prostate cancer. The imaging of 212Pb,... Read moreGeneral/Advanced Imaging
view channelBone Density Test Uses Existing CT Images to Predict Fractures
Osteoporotic fractures are not only devastating and deadly, especially hip fractures, but also impose significant costs. They rank among the top chronic diseases in terms of disability-adjusted life years... Read more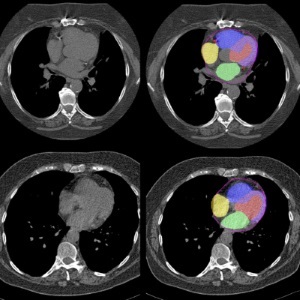
AI Predicts Cardiac Risk and Mortality from Routine Chest CT Scans
Heart disease remains the leading cause of death and is largely preventable, yet many individuals are unaware of their risk until it becomes severe. Early detection through screening can reveal heart issues,... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more