Company Developing Cerenkov Luminescence Imaging Technologies Raises Funding for Innovative Products
|
By MedImaging International staff writers Posted on 13 Jan 2015 |
A developer of novel imaging technologies has received GBP 2 million in funding to help bring its lead products to market in 2015.
The products, currently in clinical trials at Guy's Hospital (Great Maze Pond, London, United Kingdom) can help surgeons detect cancer in real-time during surgery using optical detection of Positron Emission Tomography (PET) tracer, and will reduce the high rate of repeat operations especially in breast and prostate cancer patients.
In the United Kingdom one in four women that have surgery for early-stage breast cancer need a second operation. Using current techniques surgeons are not able to accurately detect tiny cancerous deposits during an operation. The undetected cancerous tissues remain and need to be removed in one or more follow-up operations.
The LightPath specimen analyzer and the EnLight molecular imaging fiberscope were developed by Lightpoint Medical (London, United Kingdom).
Dr. David Tuch, CEO of Lightpoint Medica said: “The level of investment we have raised reflects the real clinical demand for this technology, and therefore its enormous market potential. The current rate of re-operations comes with a high cost to patient well-being and healthcare budgets, and we are very pleased that the funding will help bring our products to people who need them.”
Related Links:
Guy's Hospital
Lightpoint Medical
The products, currently in clinical trials at Guy's Hospital (Great Maze Pond, London, United Kingdom) can help surgeons detect cancer in real-time during surgery using optical detection of Positron Emission Tomography (PET) tracer, and will reduce the high rate of repeat operations especially in breast and prostate cancer patients.
In the United Kingdom one in four women that have surgery for early-stage breast cancer need a second operation. Using current techniques surgeons are not able to accurately detect tiny cancerous deposits during an operation. The undetected cancerous tissues remain and need to be removed in one or more follow-up operations.
The LightPath specimen analyzer and the EnLight molecular imaging fiberscope were developed by Lightpoint Medical (London, United Kingdom).
Dr. David Tuch, CEO of Lightpoint Medica said: “The level of investment we have raised reflects the real clinical demand for this technology, and therefore its enormous market potential. The current rate of re-operations comes with a high cost to patient well-being and healthcare budgets, and we are very pleased that the funding will help bring our products to people who need them.”
Related Links:
Guy's Hospital
Lightpoint Medical
Latest Industry News News
- Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
- Bayer and Google Partner on New AI Product for Radiologists
- Samsung and Bracco Enter Into New Diagnostic Ultrasound Technology Agreement
- IBA Acquires Radcal to Expand Medical Imaging Quality Assurance Offering
- International Societies Suggest Key Considerations for AI Radiology Tools
- Samsung's X-Ray Devices to Be Powered by Lunit AI Solutions for Advanced Chest Screening
- Canon Medical and Olympus Collaborate on Endoscopic Ultrasound Systems
- GE HealthCare Acquires AI Imaging Analysis Company MIM Software
- First Ever International Criteria Lays Foundation for Improved Diagnostic Imaging of Brain Tumors
- RSNA Unveils 10 Most Cited Radiology Studies of 2023
- RSNA 2023 Technical Exhibits to Offer Innovations in AI, 3D Printing and More
- AI Medical Imaging Products to Increase Five-Fold by 2035, Finds Study
- RSNA 2023 Technical Exhibits to Highlight Latest Medical Imaging Innovations
- AI-Powered Technologies to Aid Interpretation of X-Ray and MRI Images for Improved Disease Diagnosis
- Hologic and Bayer Partner to Improve Mammography Imaging
- Global Fixed and Mobile C-Arms Market Driven by Increasing Surgical Procedures
Channels
Radiography
view channel
Novel Breast Imaging System Proves As Effective As Mammography
Breast cancer remains the most frequently diagnosed cancer among women. It is projected that one in eight women will be diagnosed with breast cancer during her lifetime, and one in 42 women who turn 50... Read more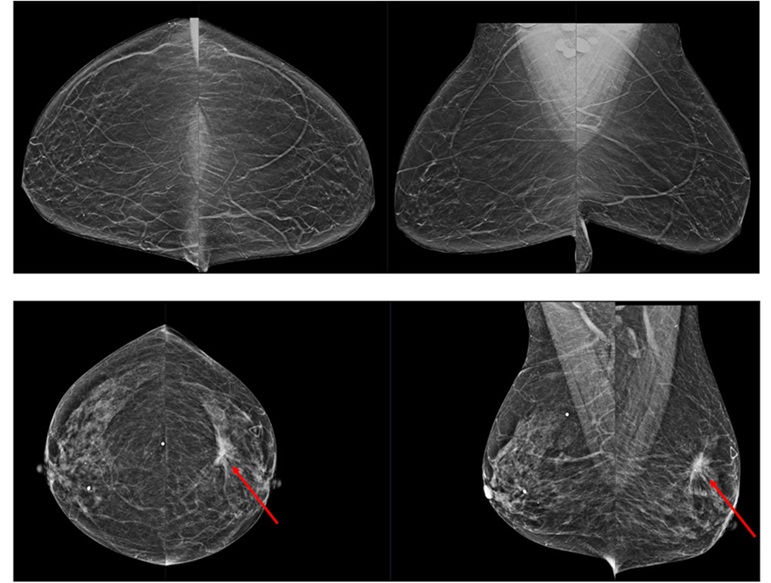
AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
Radiologists typically detect one case of cancer for every 200 mammograms reviewed. However, these evaluations often result in false positives, leading to unnecessary patient recalls for additional testing,... Read moreMRI
view channel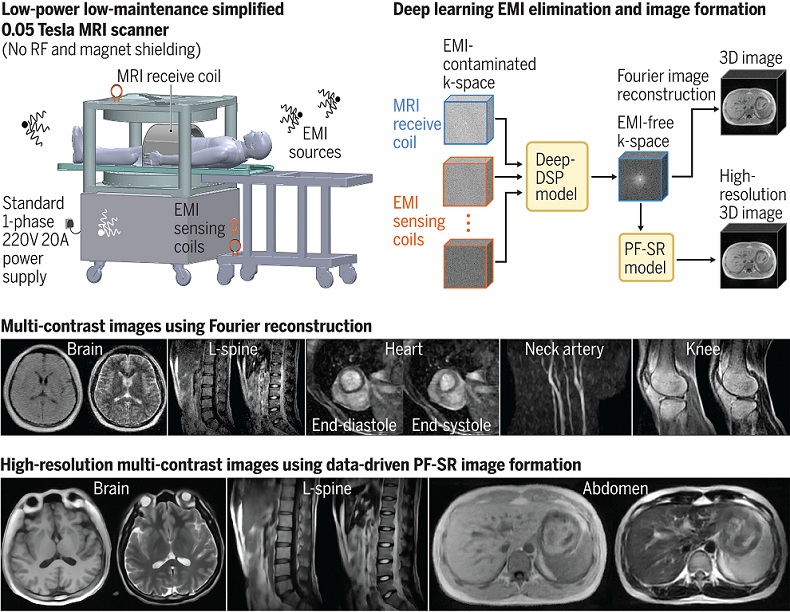
Low-Cost Whole-Body MRI Device Combined with AI Generates High-Quality Results
Magnetic Resonance Imaging (MRI) has significantly transformed healthcare, providing a noninvasive, radiation-free method for detailed imaging. It is especially promising for the future of medical diagnosis... Read more
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read moreUltrasound
view channel.jpg)
Diagnostic System Automatically Analyzes TTE Images to Identify Congenital Heart Disease
Congenital heart disease (CHD) is one of the most prevalent congenital anomalies worldwide, presenting substantial health and financial challenges for affected patients. Early detection and treatment of... Read more
Super-Resolution Imaging Technique Could Improve Evaluation of Cardiac Conditions
The heart depends on efficient blood circulation to pump blood throughout the body, delivering oxygen to tissues and removing carbon dioxide and waste. Yet, when heart vessels are damaged, it can disrupt... Read more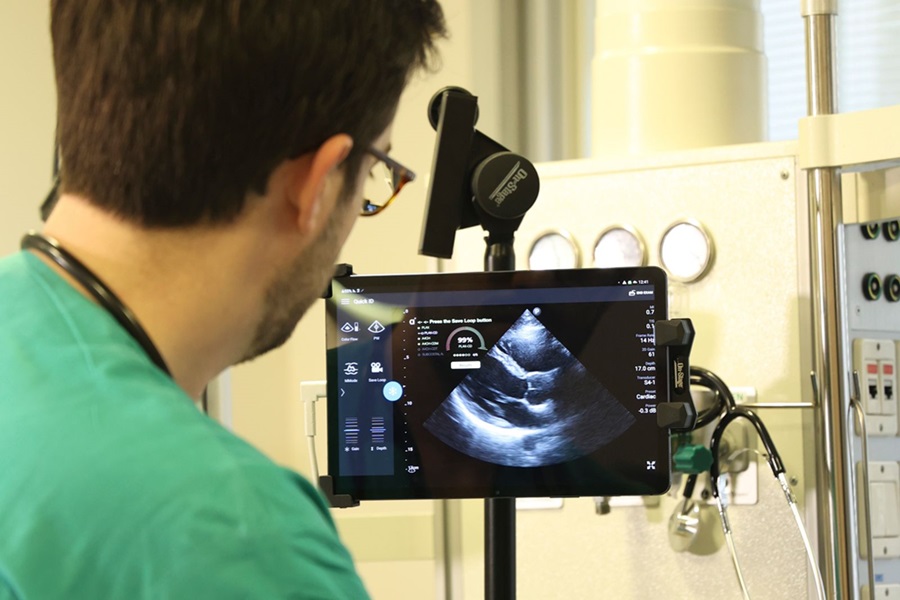
First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity
Ultrasound scans are essential for identifying and diagnosing various medical conditions, but often, patients must wait weeks or months for results due to a shortage of qualified medical professionals... Read moreNuclear Medicine
view channel
New PET Biomarker Predicts Success of Immune Checkpoint Blockade Therapy
Immunotherapies, such as immune checkpoint blockade (ICB), have shown promising clinical results in treating melanoma, non-small cell lung cancer, and other tumor types. However, the effectiveness of these... Read moreNew PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
Clear cell renal cell cancer (ccRCC) represents 70-80% of renal cell carcinoma cases. While localized disease can be effectively treated with surgery and ablative therapies, one-third of patients either... Read more
New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
Imaging inflammation using traditional radiological techniques presents significant challenges, including radiation exposure, poor image quality, high costs, and invasive procedures. Now, new contrast... Read more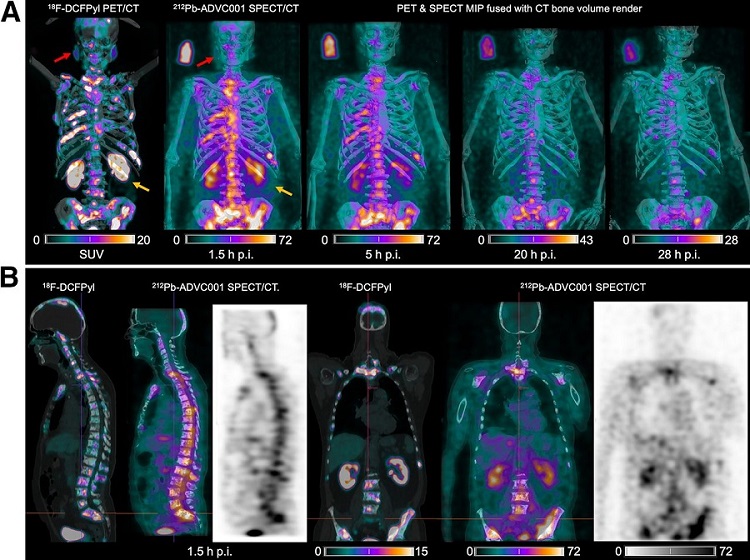
New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
The development of lead-212 (212Pb)-PSMA–based targeted alpha therapy (TAT) is garnering significant interest in treating patients with metastatic castration-resistant prostate cancer. The imaging of 212Pb,... Read moreGeneral/Advanced Imaging
view channelBone Density Test Uses Existing CT Images to Predict Fractures
Osteoporotic fractures are not only devastating and deadly, especially hip fractures, but also impose significant costs. They rank among the top chronic diseases in terms of disability-adjusted life years... Read more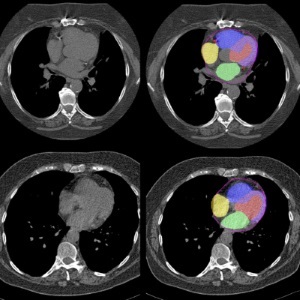
AI Predicts Cardiac Risk and Mortality from Routine Chest CT Scans
Heart disease remains the leading cause of death and is largely preventable, yet many individuals are unaware of their risk until it becomes severe. Early detection through screening can reveal heart issues,... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more