X-Rays Provide New Clues into Muscle Contraction Mechanics
|
By MedImaging International staff writers Posted on 26 Apr 2011 |
In a noted experiment first performed more than 220 years ago, Italian physician Luigi Galvani discovered that the muscles of a frog's leg twitch when an electric voltage is applied. An international group of scientists from Italy, the United Kingdom, and France has now brought this classic textbook case into the nanoscience era. They used a powerful new synchrotron X-ray method to track, for the first time, at the molecular scale how muscle proteins change form and structure inside an intact and contracting muscle cell.
The study's findings are published in the April 11, 2011, issue of the journal Proceedings of the [US] National Academy of Sciences (PNAS). The team included scientists from Università di Firenze (Italy), King's College London (UK) and the European Synchrotron Radiation Facility (ESRF; Grenoble (France).
A muscle cell contains two sets of filaments composed of the proteins actin and myosin, respectively. Muscles contract as a result of the relative sliding of these filaments. When the brain sends a nerve signal to activate a muscle, the electrical signal is transmitted to the muscle cell. This initiates a chain of events inside the muscle cell that ultimately leads to alterations in the structure of the myosin and actin filaments.
But what precisely occurs during this process at the molecular level? "As we need muscles for locomotion, breathing and body posture, and for the contraction of the heart, understanding of these mechanisms has broad significance in biology and medicine,” said Dr. Malcolm Irving, from King's College London, and coauthor of the study.
"Studying a single intact and contracting muscle cell at the molecular scale in milliseconds became possible thanks to a new technique called X-ray interferometry based on low angle diffraction. This requires the extremely intense and narrow beam of X-rays provided by the ESRF,” added Dr. Theyencheri Narayanan from the ESRF, another coauthor of the paper.
The results of the research revealed the conformation of the head domains of myosin--the molecular motors that drive filament sliding--in resting muscle, and show that the movements of the myosin motors following muscle activation are much slower than the structural changes in the actin filaments. The different timings of the structural changes reveal the signaling pathway between the actin and myosin filaments in muscle, shedding new light on the mechanism of muscle regulation.
"We were observing quite rapid biological processes, in the order of milliseconds, along with minuscule structural changes, typically 10 nm or less. A human hair is ten-thousand times thicker. To integrate the mechanical and X-ray diffraction methods that are required to resolve the combination of these extremes is a real experimental challenge,” explained Dr. Vincenzo Lombardi, from the University of Florence.
Although the study has no immediate clinical application, its longer-term impact may be in the field of heart disease, in which these essential signaling processes are not working optimally in the heart muscle.
Existing agents to treat heart failure by modulating this signaling pathway are far from ideal. "To develop better drugs for heart failure, it's likely that a better understanding of the molecular mechanisms of muscle regulation is needed, and that may be the long-term contribution of our experiments,” concluded Dr. Irving.
Related Links:
Università di Firenze
King's College London
European Synchrotron Radiation Facility
The study's findings are published in the April 11, 2011, issue of the journal Proceedings of the [US] National Academy of Sciences (PNAS). The team included scientists from Università di Firenze (Italy), King's College London (UK) and the European Synchrotron Radiation Facility (ESRF; Grenoble (France).
A muscle cell contains two sets of filaments composed of the proteins actin and myosin, respectively. Muscles contract as a result of the relative sliding of these filaments. When the brain sends a nerve signal to activate a muscle, the electrical signal is transmitted to the muscle cell. This initiates a chain of events inside the muscle cell that ultimately leads to alterations in the structure of the myosin and actin filaments.
But what precisely occurs during this process at the molecular level? "As we need muscles for locomotion, breathing and body posture, and for the contraction of the heart, understanding of these mechanisms has broad significance in biology and medicine,” said Dr. Malcolm Irving, from King's College London, and coauthor of the study.
"Studying a single intact and contracting muscle cell at the molecular scale in milliseconds became possible thanks to a new technique called X-ray interferometry based on low angle diffraction. This requires the extremely intense and narrow beam of X-rays provided by the ESRF,” added Dr. Theyencheri Narayanan from the ESRF, another coauthor of the paper.
The results of the research revealed the conformation of the head domains of myosin--the molecular motors that drive filament sliding--in resting muscle, and show that the movements of the myosin motors following muscle activation are much slower than the structural changes in the actin filaments. The different timings of the structural changes reveal the signaling pathway between the actin and myosin filaments in muscle, shedding new light on the mechanism of muscle regulation.
"We were observing quite rapid biological processes, in the order of milliseconds, along with minuscule structural changes, typically 10 nm or less. A human hair is ten-thousand times thicker. To integrate the mechanical and X-ray diffraction methods that are required to resolve the combination of these extremes is a real experimental challenge,” explained Dr. Vincenzo Lombardi, from the University of Florence.
Although the study has no immediate clinical application, its longer-term impact may be in the field of heart disease, in which these essential signaling processes are not working optimally in the heart muscle.
Existing agents to treat heart failure by modulating this signaling pathway are far from ideal. "To develop better drugs for heart failure, it's likely that a better understanding of the molecular mechanisms of muscle regulation is needed, and that may be the long-term contribution of our experiments,” concluded Dr. Irving.
Related Links:
Università di Firenze
King's College London
European Synchrotron Radiation Facility
Latest Radiography News
- Novel Breast Imaging System Proves As Effective As Mammography
- AI Assistance Improves Breast-Cancer Screening by Reducing False Positives
- AI Could Boost Clinical Adoption of Chest DDR
- 3D Mammography Almost Halves Breast Cancer Incidence between Two Screening Tests
- AI Model Predicts 5-Year Breast Cancer Risk from Mammograms
- Deep Learning Framework Detects Fractures in X-Ray Images With 99% Accuracy
- Direct AI-Based Medical X-Ray Imaging System a Paradigm-Shift from Conventional DR and CT
- Chest X-Ray AI Solution Automatically Identifies, Categorizes and Highlights Suspicious Areas
- AI Diagnoses Wrist Fractures As Well As Radiologists
- Annual Mammography Beginning At 40 Cuts Breast Cancer Mortality By 42%
- 3D Human GPS Powered By Light Paves Way for Radiation-Free Minimally-Invasive Surgery
- Novel AI Technology to Revolutionize Cancer Detection in Dense Breasts
- AI Solution Provides Radiologists with 'Second Pair' Of Eyes to Detect Breast Cancers
- AI Helps General Radiologists Achieve Specialist-Level Performance in Interpreting Mammograms
- Novel Imaging Technique Could Transform Breast Cancer Detection
- Computer Program Combines AI and Heat-Imaging Technology for Early Breast Cancer Detection
Channels
MRI
view channel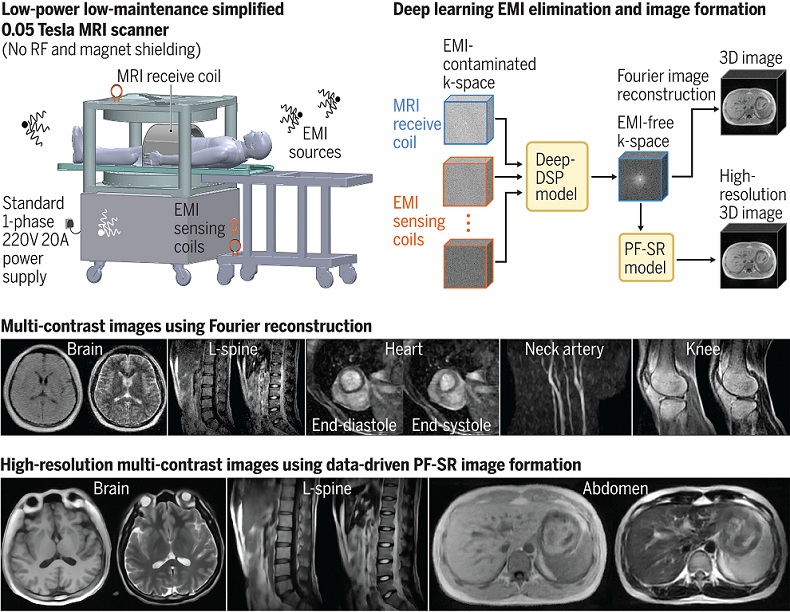
Low-Cost Whole-Body MRI Device Combined with AI Generates High-Quality Results
Magnetic Resonance Imaging (MRI) has significantly transformed healthcare, providing a noninvasive, radiation-free method for detailed imaging. It is especially promising for the future of medical diagnosis... Read more
World's First Whole-Body Ultra-High Field MRI Officially Comes To Market
The world's first whole-body ultra-high field (UHF) MRI has officially come to market, marking a remarkable advancement in diagnostic radiology. United Imaging (Shanghai, China) has secured clearance from the U.... Read moreUltrasound
view channel.jpg)
Diagnostic System Automatically Analyzes TTE Images to Identify Congenital Heart Disease
Congenital heart disease (CHD) is one of the most prevalent congenital anomalies worldwide, presenting substantial health and financial challenges for affected patients. Early detection and treatment of... Read more
Super-Resolution Imaging Technique Could Improve Evaluation of Cardiac Conditions
The heart depends on efficient blood circulation to pump blood throughout the body, delivering oxygen to tissues and removing carbon dioxide and waste. Yet, when heart vessels are damaged, it can disrupt... Read more
First AI-Powered POC Ultrasound Diagnostic Solution Helps Prioritize Cases Based On Severity
Ultrasound scans are essential for identifying and diagnosing various medical conditions, but often, patients must wait weeks or months for results due to a shortage of qualified medical professionals... Read moreNuclear Medicine
view channel
New PET Biomarker Predicts Success of Immune Checkpoint Blockade Therapy
Immunotherapies, such as immune checkpoint blockade (ICB), have shown promising clinical results in treating melanoma, non-small cell lung cancer, and other tumor types. However, the effectiveness of these... Read moreNew PET Agent Rapidly and Accurately Visualizes Lesions in Clear Cell Renal Cell Carcinoma Patients
Clear cell renal cell cancer (ccRCC) represents 70-80% of renal cell carcinoma cases. While localized disease can be effectively treated with surgery and ablative therapies, one-third of patients either... Read more
New Imaging Technique Monitors Inflammation Disorders without Radiation Exposure
Imaging inflammation using traditional radiological techniques presents significant challenges, including radiation exposure, poor image quality, high costs, and invasive procedures. Now, new contrast... Read more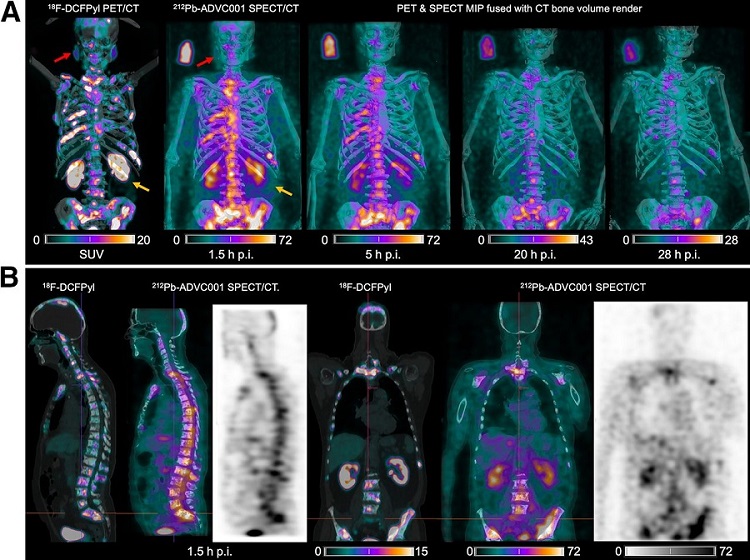
New SPECT/CT Technique Could Change Imaging Practices and Increase Patient Access
The development of lead-212 (212Pb)-PSMA–based targeted alpha therapy (TAT) is garnering significant interest in treating patients with metastatic castration-resistant prostate cancer. The imaging of 212Pb,... Read moreGeneral/Advanced Imaging
view channelBone Density Test Uses Existing CT Images to Predict Fractures
Osteoporotic fractures are not only devastating and deadly, especially hip fractures, but also impose significant costs. They rank among the top chronic diseases in terms of disability-adjusted life years... Read more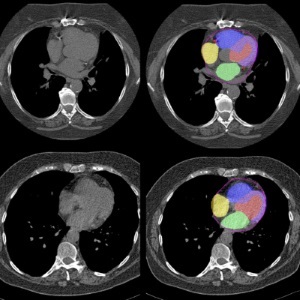
AI Predicts Cardiac Risk and Mortality from Routine Chest CT Scans
Heart disease remains the leading cause of death and is largely preventable, yet many individuals are unaware of their risk until it becomes severe. Early detection through screening can reveal heart issues,... Read moreImaging IT
view channel
New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
Medical imaging is a critical tool used to diagnose patients, and there are billions of medical images scanned globally each year. Imaging data accounts for about 90% of all healthcare data1 and, until... Read more
Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
The global artificial intelligence (AI) in medical diagnostics market is expanding with early disease detection being one of its key applications and image recognition becoming a compelling consumer proposition... Read moreIndustry News
view channel
Hologic Acquires UK-Based Breast Surgical Guidance Company Endomagnetics Ltd.
Hologic, Inc. (Marlborough, MA, USA) has entered into a definitive agreement to acquire Endomagnetics Ltd. (Cambridge, UK), a privately held developer of breast cancer surgery technologies, for approximately... Read more
Bayer and Google Partner on New AI Product for Radiologists
Medical imaging data comprises around 90% of all healthcare data, and it is a highly complex and rich clinical data modality and serves as a vital tool for diagnosing patients. Each year, billions of medical... Read more