Ultrasound System Designed for Wide Range of Applications
By MedImaging staff writers
Posted on 25 Jun 2008
A new color ultrasound imaging system is designed to have extensive applications in abdominal, obstetrics/gynecology, endovaginal, cardiac, small parts, and pediatric markets. The system is ideally suited for hospitals, doctors' offices and veterinary clinics seeking to replace black and white ultrasound imaging systems.Posted on 25 Jun 2008
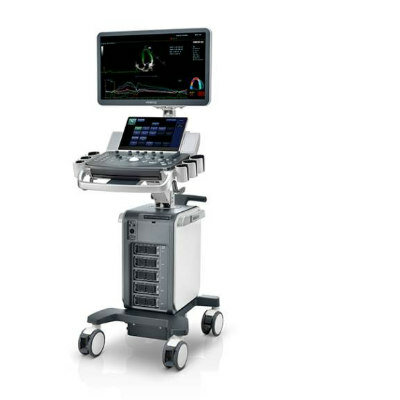
Mindray Medical International, Ltd. (Shenzhen, China), a developer, manufacturer, and marketer of medical devices worldwide, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its DC-3 color ultrasound imaging system. The clearance comes in addition to China State Food and Drug Administration (SFDA) approval and CE Marking granted earlier in 2008 for the sale of the DC-3 in Chinese and European markets, respectively.
"The compact DC-3 combines outstanding 2D [two-dimensional] imaging and exceptional color Doppler performance,” said Li Xiting, Mindray's president and co-chief executive officer. "With a superior price-to-performance ratio, we are confident that this versatile product will meet the diverse needs and requirements of hospitals, doctors' offices and veterinary clinics around the world.”
The DC-3 is customized to expand Mindray's customer base in low-end markets and complements the company's FDA-approved DC-6 and portable M5 ultrasound imaging systems, which are targeted for middle and high-end markets and received FDA 510(k) clearance in September 2006 and March 2008, respectively. The company currently offers over 10 medical imaging products of which five have received FDA 510(k) clearance.
Mindray has to date received FDA 510(k) clearance for a total of 16 products, covering patient monitoring and life-support products, in-vitro diagnostic products, and medical imaging systems.
Related Links:
Mindray Medical International