New Consensus Standardizes Ultrasound-Based Fatty Liver Assessment
Posted on 23 Mar 2026
Metabolic dysfunction-associated steatotic liver disease (MASLD) is rising along with obesity and diabetes, making accurate, scalable measurement of hepatic fat a clinical priority. Biopsy is invasive and magnetic resonance methods remain costly and limited in access. This gap hampers screening, risk stratification, and therapy monitoring across diverse settings. To help address this challenge, investigators have produced a clinical expert consensus on ultrasound-derived fat fraction (UDFF) to standardize noninvasive assessment of liver steatosis.
The consensus was developed by a team from Fudan University (Fudan, China) and published online in Portal Hypertension & Cirrhosis on March 6, 2026. UDFF is a quantitative ultrasound technique that expresses liver fat content as a percentage, aiming to combine wide availability with objective measurement. The document positions UDFF as a practical solution where liver biopsy and magnetic resonance imaging–derived proton density fat fraction (MRI-PDFF) are impractical.
The authors synthesized evidence from prospective multicenter trials, biopsy-based validation studies, MRI-PDFF–referenced analyses, pediatric cohorts, and studies in special populations. They compared UDFF with conventional ultrasound scoring systems, the controlled attenuation parameter, and serological indices such as the fatty liver index and hepatic steatosis index. Diagnostic performance was evaluated using correlation coefficients, intraclass correlation coefficients (ICCs), area under the receiver operating characteristic curve (AUC), and Bland–Altman analyses.
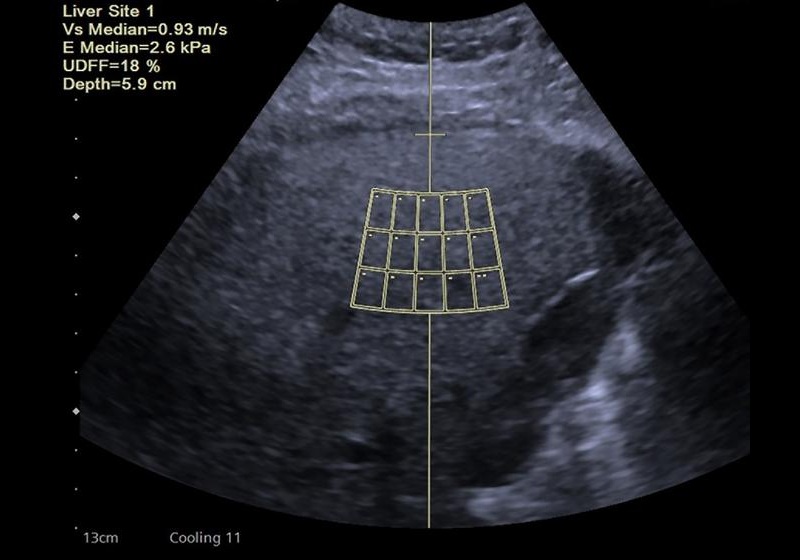
Across studies, UDFF demonstrated excellent reliability, with intra- and inter-operator ICCs of at least 0.94. UDFF correlated strongly with histological steatosis grades and even more strongly with MRI-PDFF. In multicenter analyses, AUC values commonly exceeded 0.90. A meta-analysis of 1,150 patients reported pooled sensitivity of 90.4%, specificity of 83.8%, and a summary AUC of 0.93, with low heterogeneity. UDFF frequently outperformed conventional ultrasound grading, controlled attenuation parameter, and several serological indices.
Provisional diagnostic thresholds were proposed from the largest available dataset: 8% for at least S1, 14% for at least S2, and 20% for S3 steatosis. A dual-threshold strategy using rule-in and rule-out cutoffs reduced indeterminate classifications, particularly in individuals with higher body mass index. Evidence from pediatric patients and those with comorbidities—including viral hepatitis, Wilson’s disease, and polycystic ovary syndrome—suggested consistent performance across diverse clinical contexts.
The consensus highlights UDFF as a stable, quantitative, and noninvasive method for assessing hepatic fat content, with standardized quality control criteria and preliminary cutoffs to enhance usability. It emphasizes cost-effectiveness and operational convenience, presenting UDFF as a practical alternative to biopsy and advanced magnetic resonance techniques in many care environments.
The authors note limitations related to heterogeneous reference standards and data gaps in some subgroups, and call for large, multicenter studies with unified benchmarks to refine thresholds and clarify UDFF’s role in identifying high-risk metabolic dysfunction-associated steatohepatitis, especially with significant fibrosis.






 Guided Devices.jpg)