FDA Clears 3D Total Knee Arthroplasty Surgical Planning Software
By Andrew Deutsch
Posted on 16 Nov 2016
The US FDA has cleared a new 3D total knee arthroplasty surgical planning software that is part of a portfolio of apps for online weight-bearing 3D surgical planning for common orthopedic treatments.Posted on 16 Nov 2016
The online 3D planning software uses weight-bearing 3D images and other data from a 2D/3D orthopedic imaging system and provides surgical planning for total knee arthroplasty procedures.
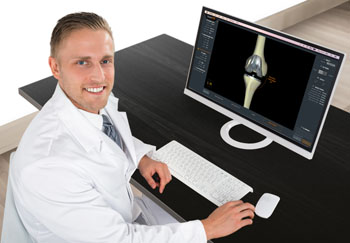
Image: A clinician using the KneeEOS total knee arthroplasty surgical planning software (Photo courtesy of EOS Imaging).
The EOS Imaging (Paris, France) KneeEOS online software planning tool received 510(k) clearance from the US Food and Drug Administration (FDA; Silver Spring, MD USA) for marketing in the US.
KneeEOS is designed for use with the EOS Imaging 2D/3D orthopedic imaging system and is the part of a portfolio of EOSapps for common orthopedic surgeries. KneeEOS creates an initial proposal for size and position of implants to help surgeons prepare and optimize the alignment of the implants in 3D, and also displays relevant clinical data in real time.
EOSapps use 2D images, and 3D digital anatomical datasets taken using the EOS imaging system for diagnosis, pre-operative surgical planning, post-operative assessment of the patient, and for follow-up imaging.
Marie Meynadier, EOS Imaging CEO, said, “The kneeEOS FDA clearance is an important milestone that allows us to offer our full set of software solutions to the U.S. market. It will support the growing adoption of EOS images, 3D models and patient-specific datasets at each step of the care continuum, helping healthcare providers bring the value of personalized treatments to their patients without the high dose and cost of CT imaging.”
Related Links:
EOS Imaging
US Food and Drug Administration










 Guided Devices.jpg)